Clinical and anatomic description of patients with arteriovenous malformation treated with endovascular therapy in a Mexican population
Article information
Abstract
Objective
Arteriovenous malformations (AVMs) are benign congenital lesions. The estimated prevalence is 10-18 per 100,000 individuals, with an incidence rate of 1.1-1.4 per 100,000 person-year; only 12% of AVMs present symptoms during life. It is important to study different characteristics associated with AVMs because these patients require multidisciplinary treatment.
Methods
A descriptive, observational, longitudinal, and retrospective study was carried out from January 15, 2016 to October 19, 2021. Convenience sampling was used on patients diagnosed with arteriovenous malformation. Sociodemographic data was obtained, such as: sex, age, site, predominant symptoms, clinical debut, type of malformation, data associated with post therapy evolution, type of embolizing agent used, associated complications, Rankin scale, and death.
Results
Data from 535 procedures was collected, we calculated an incidence of 4.4 cases per million inhabitants. Ninety procedures in 56 patients that used endovascular therapy were obtained with a female-male ratio of 0.75 and an age distribution of 35±14 (SD). A 36.3±11.5 (SD) mm diameter was registered. There was a 33% success rate for endovascular surgery.
Conclusions
The multidisciplinary treatment of AVMs is made up of three pillar techniques: microsurgical resection, embolization, and radio-neurosurgery. It is extremely important to make an appropriate decision, with an emphasis on achieving better functional outcomes for the patient. Although neurological endovascular therapy was initially used as an adjuvant treatment for neurosurgery and radio-neurosurgery, it has been used more and more frequently as the first line of treatment.
INTRODUCTION
Intracranial arteriovenous malformations (AVMs) are lesions made up of arteries and veins without any intervening capillaries. In other words, they are made up of afferent branches that lead to a bed of gnarled vessels with no intervening capillaries. They are found in the brain parenchyma and alternate with efferent branches that drain the blood that feed them. Therefore, they are defined as a set of dilated arteries that connect to a bed of vessels that are drained by arterialized veins, forming a high-flow, low-resistance short circuit between the arterial and venous systems [25,35,37]. It is considered a benign vascular entity of exclusively congenital origin that develops in the prenatal period. They are associated with hereditary hemorrhagic telangiectasia (HHT), Wyburn-Mason syndrome, Osler-Weber-Rendu disease, and Sturge-Weber syndrome [16,25].
The estimated prevalence is 10–18 per 100,000 individuals, and the incidence rate is 1.1–1.4 per 100,000 individuals/year [16]. Only 12% of AVMs are symptomatic [16,36]. Current studies have shown that inflammation is associated with the genesis of sporadic AVMs as well as their growth and rupture [16]. On the other hand, AVMs can cause neurological morbidity because of their loco-regional effects in adjacent areas of the brain. This is due to the high flow of the arteriovenous shunt within the AVM that can divert blood from the surrounding capillary network of the normal parenchyma, triggering a state of hypoperfusion that later leads to the dilation of the capillary network adjacent to the nidus and recruitment of leptomeningeal collaterals [6].
Intracranial hemorrhage is the most common symptomatic manifestation of an AVM. However, the prognosis of a hemorrhage secondary to AVM is associated with a better prognosis than that of a spontaneous primary hemorrhage. The overall risk of bleeding from an untreated, unruptured AVM is estimated to be 1% to 3% per year [3,5]. This risk is higher among previously ruptured AVMs compared to unruptured AVMs, especially during the first year after the initial hemorrhage [22].
The increasing ease with which brain imaging studies are now obtained, such as contrast-enhanced tomography and magnetic resonance angiography, has made it possible to detect more and more AVMs incidentally. However, the gold standard for its study is diagnostic cerebral angiography [12].
The goal of AVM treatment is the complete obliteration of the nidus and all arteriovenous shunts since there is consensus that partial obliteration of AVMs does not reduce the risk of bleeding [36]. Therefore, there are three treatment modalities for AVMs: microsurgical resection, stereotaxic radiosurgery, and endovascular embolization. Microsurgery has been associated with high levels of obliteration, which is why it is commonly used as first-line treatment. There are adjuvant elements to surgery that improve its performance, such as advanced preoperative neuroimaging, preoperative embolization, frameless stereotactic neuronavigation, intraoperative vascular imaging, intraoperative electrophysiological monitoring, and mapping [15,21].
Stereotactic radiosurgery causes vascular damage that gradually leads to the sclerosis of blood vessels and eventual obliteration of the AVM over a period of 2 to 3 years on average. Its use is more suitable for small or medium-sized AVMs (volume 12 cm [25] or diameter 3 cm) located in deep or eloquent brain regions [15,21,26,33]. Endovascular embolization is usually performed as adjuvant therapy before microsurgical resection or radiosurgery as part of the multimodal management of AVMs. Embolization can be used as a curative treatment or palliative treatment in which flow is reduced to decrease symptoms that are potentially caused by local venous hypertension or vascular steal [27,30,38].
These types of patients require multidisciplinary management (neurosurgery, radio-neurosurgery, and endovascular therapy), to individualize each case, it is important to study the different characteristics associated with AVMs, both clinically and socially. This helps set the standard for the best management plan according to the AVM classification and the patient’s context, coupled with the advantage of new technologies in endovascular therapy such as catheters, guides, devices, and other materials for treatment of vascular pathologies other than AVMs (cerebral vascular events, intracranial aneurysms, carotid disease, among others). These materials have also been useful for treatment with increasingly better results with the use of non-adhesive liquid embolic agent comprised of a biocompatible polymer dissolved in dimethyl sulfoxide (DMSO) solvent with the commercial name of PHIL®, coils, squid, Onyx, and N-butyl-cyanoacrylate (NBCA), as well as intravascular balloon assistance and the different approaches (trans-arterial vs. trans-venous) that have increased the percentage of obliteration in AVMs by 93–97% in selected cases, including those with Spetzler-Martin grade III [4,24].
The aim of this study was to analytically describe a population with intracranial arteriovenous malformations treated by an endovascular approach.
MATERIALS AND METHODS
Study design
A descriptive, observational, longitudinal, and retrospective study was carried out from January 2016 to October 2021 according to the STROBE [34] recommendations. Convenience sampling was used on patients diagnosed with arteriovenous malformation who attended the Instituto Nacional de Neurología y Neurocirugía “Dr. Manuel Velasco Suárez” (I.N.N.N). The research standards were followed in accordance with the provisions of the Declaration of Helsinki. This study was approved by the institutional review board with waiver of informed consent or exemption at our institution (approval number 100/21).
Mexican health system
There are 13 institutions in Mexico called Institutos Nacionales de Salud (National Institutes of Health), whose purpose is to offer highly specialized medical care services, training of qualified health care professionals and scientific research [10]. These institutes are part of the Mexican public health care system. The I.N.N.N. is part of these institutions and is among the most prominent regarding the treatment and study of neurological disorders [7]. The I.N.N.N. often receives patients from all over the country that are sent when their primary health care centers lack the materials and qualified professionals to deliver a proper diagnosis and treatment.
Criteria selection
Patients for this study met the following inclusion criteria: age between 10 and 90 years old, presentation of incidental intracranial arteriovenous malformation or intracranial hemorrhage secondary to rupture of the arteriovenous malformation and treated with embolization of intracranial arteriovenous malformation. Patients with diagnosis of dural fistula, cavernous angioma, and/or malformations or anomalies of venous drainage and patients with intracranial arteriovenous malformation and who had undergone microsurgical resection or radio-neurosurgery without embolization were excluded. Patients who did not have complete imaging studies (magnetic resonance imaging and diagnostic cerebral angiography) within their clinical file were eliminated.
It should be noted that the decision for the treatment of this type of patient was submitted to the AVM committee monthly and subjected to a unanimous decision by the three participating services (Vascular Neurosurgery, Neurological Endovascular Therapy, and Radio-neurosurgery).
Outcome
Patients were routinely checked up on a month, three months and six months following treatment in an out-patient program. Follow up MRA was performed prior to X-ray angiography and both were preformed within six months (on average, two months) of treatment. For practical purposes, patients were classified according to the classification described by Spetzler-Martin [32].
Modified Rankin Scale (mRS) [2,28,39] was checked at admission and six months later, and a favorable outcome was defined as an mRS ≤2. After the initial evaluation and data collection, the Lawton-Young [19] and Buffalo [8] supplementary scales were calculated.
Patients were defined as cured if complete obliteration was observed in their last imaging study (cerebral angiography with digital subtraction or brain magnetic resonance imaging with contrast).
Information collection technique:
Sociodemographic data, such as sex, age, location, predominant symptoms and/or clinical debut, and neurological deterioration, were obtained. Data collection was done manually and daily, by verification of the electronic clinical file. In the case of missing data, the notebooks of each of the hemodynamic rooms were reviewed. Once the data of patients diagnosed with intracranial arteriovenous malformation was available, the physical clinical file was reviewed, as well as the institutional imaging system, to determine the type of intracranial arteriovenous malformation, data associated with post-intervention evolution, type of embolizing agent used, associated complications, Rankin scale, and death, among others. Finally, the electronic emptying of the data obtained was done on a Microsoft Office 2016TM package spreadsheet.
Statistical analysis
All statistical analyses were performed using SPSS version 25.0 (IBM Corp., Armonk, NY, USA) with a P-value <0.05 considered statistically significant. Descriptive statistics were used for the exposure of demographic data. The results obtained are shown in means and standard deviation when the variables are quantitative and in percentages for qualitative variables.
The calculation of the accumulated incidence was made using the formula:
RESULTS
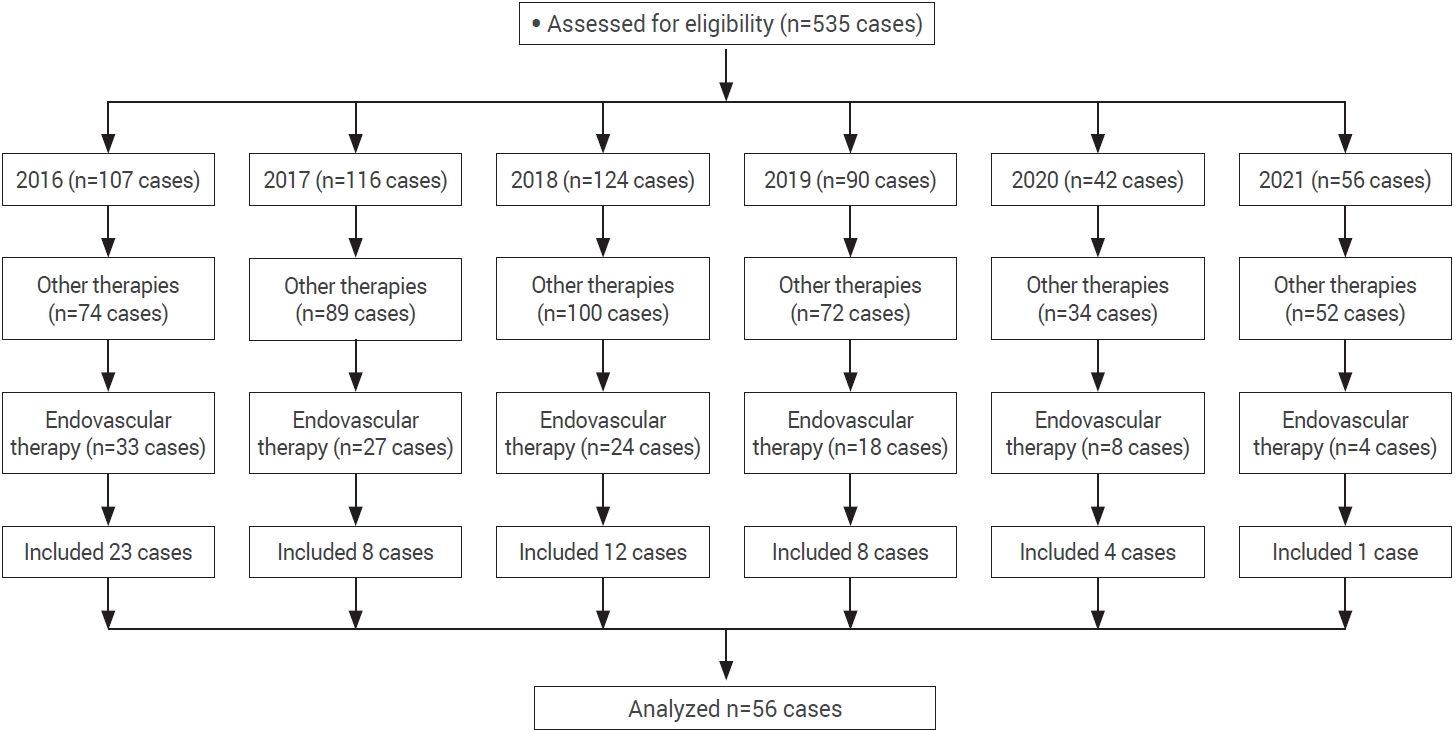
Data on 535 procedures (diagnostic and therapeutic) performed on patients diagnosed with intracranial AVM was obtained (Fig. 1). Population data provided by INEGI [14] for 2015 reported a total Mexican population of 119,938,473. By comparing this report with the results of our analysis (535 cases), we can calculate an incidence of 4.4 cases per million inhabitants.
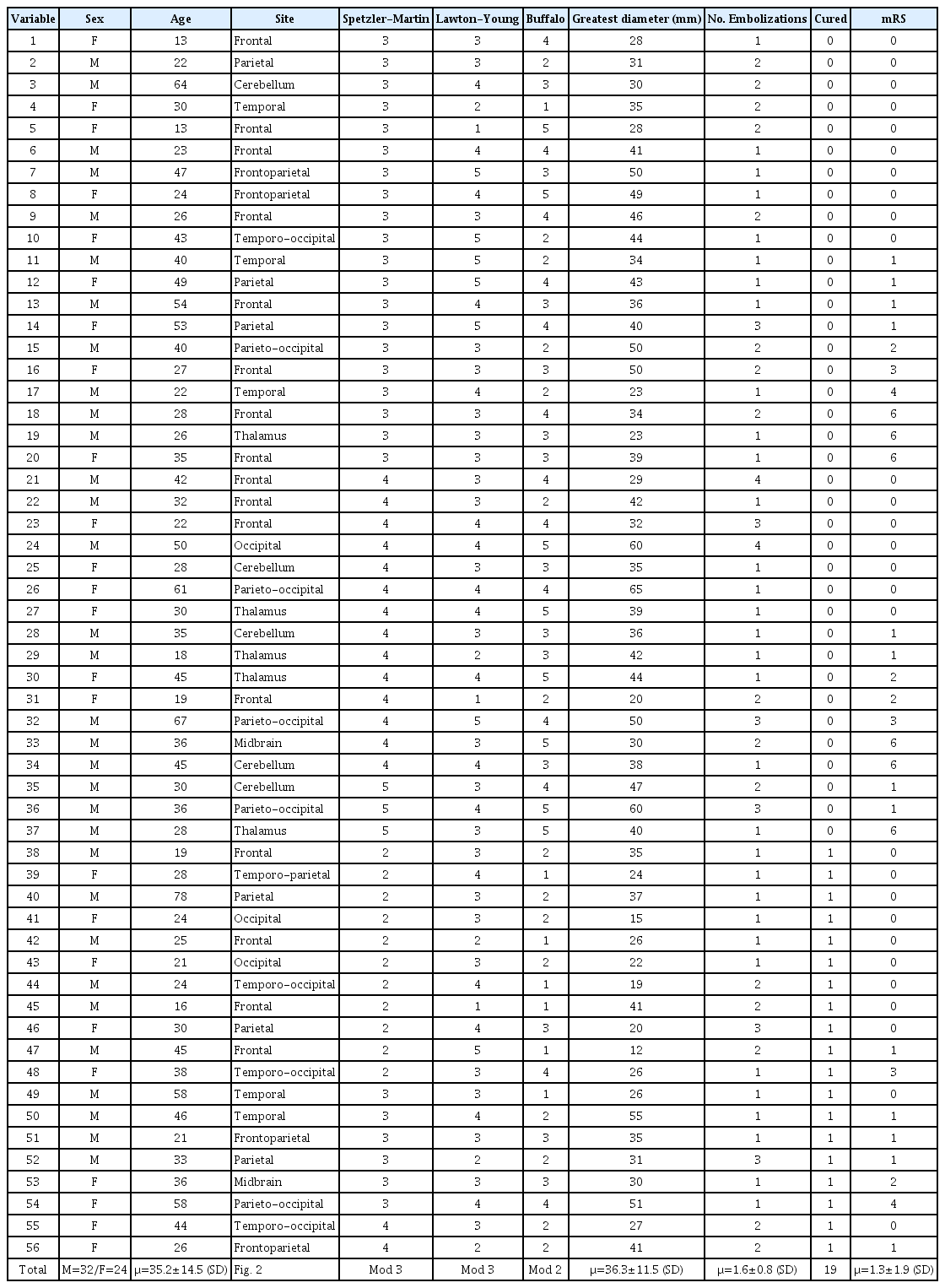
Of the total 535 cases, 421 were excluded because they were given a treatment other that embolization. Another 66 cases were eliminated because they lacked a complete clinical file and imaging studies and/or their cause of death had no correlation with AVM. Of the total number of procedures that involved endovascular embolization, 90 were performed in 56 patients (Table 1). They were mostly male (57.1%), with a male-female ratio of 0.75 and an age of 35±14 (SD) years (range: 13-78 years). Only eight cases (14.2%) with added comorbidities were found. Noteworthy comorbidities were: systemic arterial hypertension and type 2 diabetes mellitus. The rest of the results are shown in Table 2.
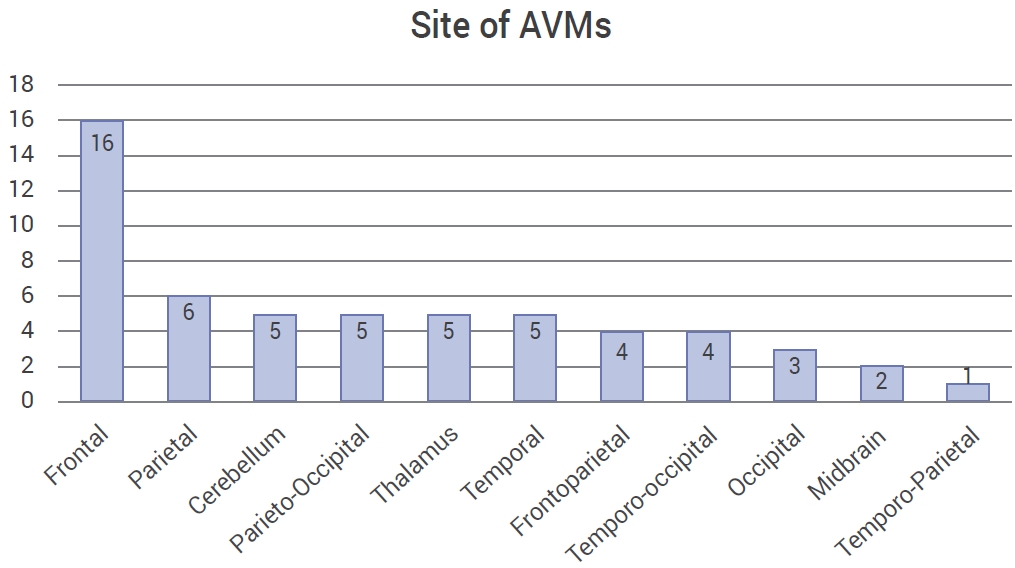
Thirty AVMs (53.6%) were identified in the right hemisphere, 22 (39.3%) on the left, and 4 were (7.1%) located in the midline. There was predominance in the frontal lobes (28.6%) and parietal lobes (10.7%) (Fig. 2). The AVMs were classified according to the Spetzler-Martin classification for arteriovenous malformations. There were no patients with a history associated to Grade I in this population. The rest of the results are shown in Table 3.
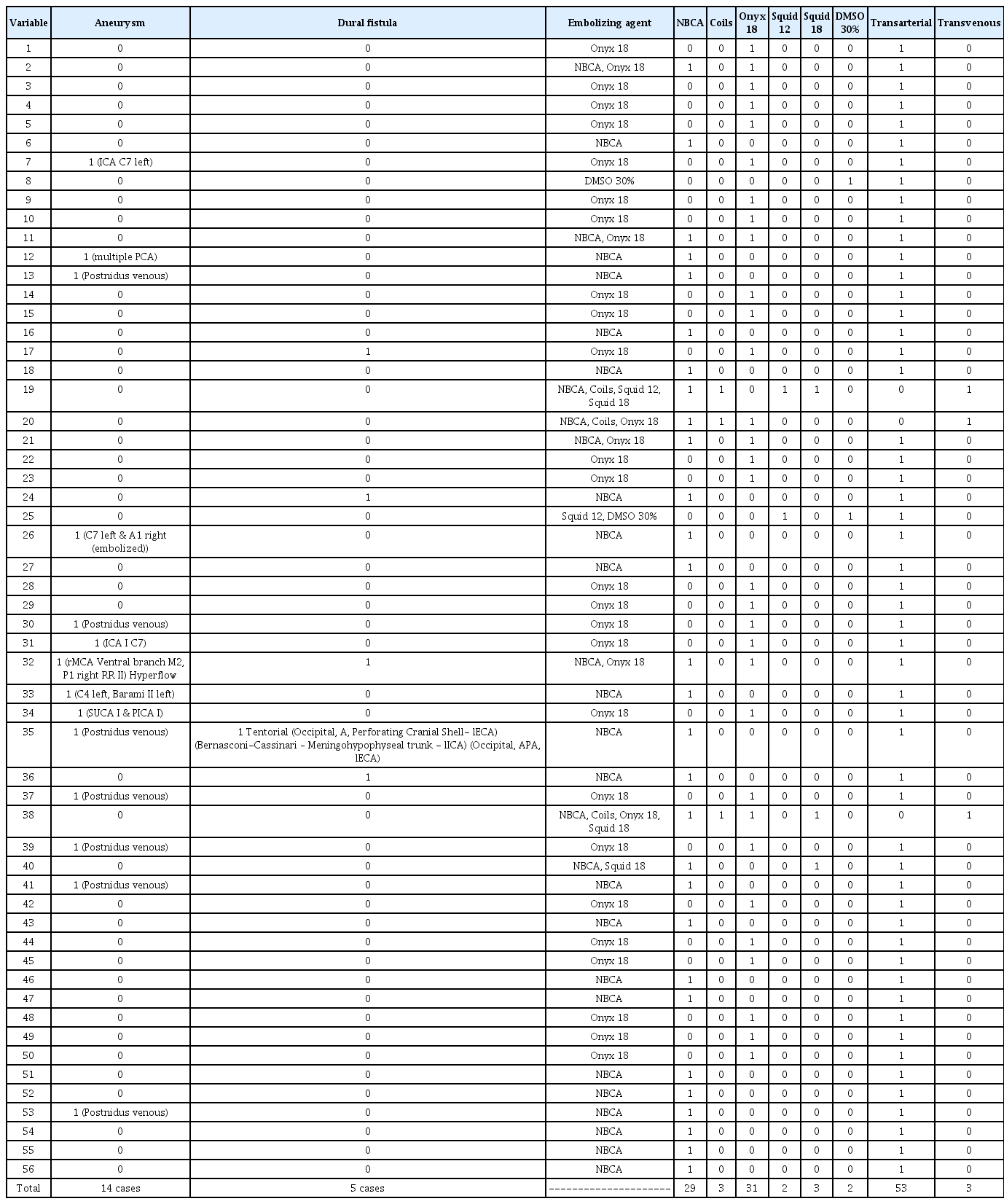
The nidus’ diameter was recorded with size ranging from 12 to 65 mm (mean 36.3±11.5 SD mm). We found 14 cases with intracranial aneurysms, of which 7 were venous (all post-nidal) and 7 arterial. In the four latter cases, they were incidental (vascular axis unrelated to the lesion) and three were classified as high flow because they were in the same axis that provides afferent vessels to the nidus, and in two of them, coil embolization of the aneurysm was performed. Five cases were found in association with a dural fistula, the most frequent being the tentorial type.
Out of the techniques available for embolization, there was a preference for the trans-arterial technique in 53 of the case (94.6%). The transvenous technique (5.3%) was performed in the rest. Ethylene-vinyl-alcohol (EVOH), commonly known as Onyx® 18 (Medtronic, Irvine, CA, USA), was used in 44.3% of cases. NBCA with the commercial name Histoacryl® (Bbraun, Melsnagen, Germany) was used in 51.8% of the cases. Squids 12 and 18 (Balt, Mont-morency, France) were used in 2 (3.6%) and 3 (5.4%) cases, respectively. The results are presented in Table 3.
Of the total population analyzed, intracranial hemorrhage was evaluated taking into consideration whether there was subarachnoid hemorrhage, intraventricular hemorrhage, and/or parenchymal hematoma, finding 16 patients (28.5%), of whom 7 had an intracranial aneurysm. Only three cases had an intracranial venous thrombosis, and 14 cases had an infarction in arterial territory. It is important to mention that four patients were drug users (marijuana, methamphetamine, cocaine, and other stimulants), and three of them had an intracranial hemorrhage and infarction. Patients with intracranial hemorrhage were subdivided according to SM as follows: I (4), II (8), IV (10), V (1), finding a preference for bleeding in those with grade IV. There was also a correlation between SM and the presence of seizures: II (9), III (20), IV (6), and V (0), showing a preference for grade III. A neurological deficit was also found in: II (2), III (19), IV (11), and V (3), with a trend again in grade III.
When the supplementary scale of Lawton-Young was applied, the following distribution was found: 3 patients with one point (5.4%), 5 patients with two points (8.9%), 24 patients with three points (42.9%), 17 patients with four points (30.4%) and 7 patients with five points (12.5%). Using the Buffalo scale, they were classified as follows: 7 patients with 1 point (12.5%), 15 patients with 2 points (26.8%), 13 patients with 3 and 4 points (23.2%) each, and 8 patients with 5 points (14.3%) (Fig. 3).
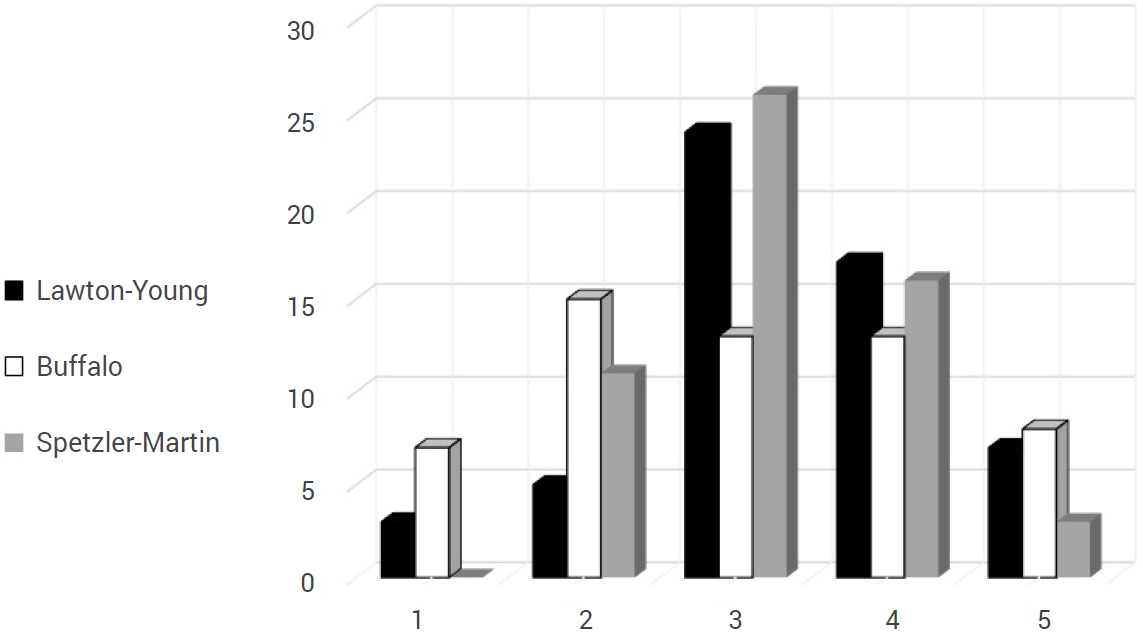
Comparative graphic between Spetzler-Martin classification and Lawton-Young scale and Buffalo score.
The following symptoms were found to be associated with AVMs: 35 patients had seizures, 25 patients had headaches, and 35 patients had a neurological deficit (alteration in the cranial nerve, motor, and/or sensory system). Most of the patients with seizures were managed by the epilepsy clinic at our institution. The drugs used to control them were the following, listed from highest to lowest frequency: levetiracetam, phenytoin, carbamazepine, acid valproic, lacosamide, oxcarbamazepine, topiramate, and amitriptyline.
Decompressive craniectomy was required in up to 6 of the patients that underwent embolization, and one patient required a craniotomy for drainage of parenchymal hematoma. Due to intraventricular hemorrhage, ventriculostomy was required on eight occasions, with five patients requiring a ventriculoperitoneal shunt.
The post-embolization functional scale that was used in these patients was the modified Rankin Scale. All cases were greater than 6 months and were categorized as satisfactory with a score between 0 and 2. This score was more frequent in MS grade III with 20 (35%) patients and Buffalo of 2 points with 14 (25%) patients.
The cure rate was 33% (19 cases). Six deaths were recorded, of which four were considered an indirect cause of endovascular treatment, with a mortality rate of 7.1%. Three of these patients underwent decompressive craniectomy. Classified with SM: three were in grade III, two were in grade IV, and one was in grade V.
The cured patients were classified according to the SM scale: 11 patients were grade II, 6 were grade III, and 2 were grade IV. Using Buffalo, they were categorized as follows: six patients with 1 point, eight patients with 2 points, two patients with 3 points, and four patients with two points.
DISCUSSION
The aim of this study was to define the clinical and anatomical characteristics of patients with a diagnosis of cerebral arteriovenous malformation who were treated by endovascular therapy. When analyzing the results, it is possible to observe that, unlike the international reports with an incidence of 1.3 cases per 100,000 patients per year determined by imaging studies [20], we calculated an incidence of 0.4 cases per 100,000 inhabitants in our population. Our findings are like those reported by Larios et al. [18], who found an incidence of 0.94 per 100,000 individuals. Larios et al. [18] and Melo Guzmán et al. [23], reported a lower age range than our study (µ=29 years and an age group 11 and 20 years, respectively). Similarly, the average age of our study was lower (35 years) than that reported in other studies, such as Pulli et al. [27] (43.22 years) and Lopes et al. [20] (44.1 years). It is worth mentioning that there were only five patients under 20 years of age, the rest were in their third decade of life, as is reported in most studies. According to Hofmeister et al. [13], when collecting their own data from five population studies with a total of 1,289 cases, they obtained 256 (19.89%) patients under 20 years of age.
In our study, the gender distribution was slightly higher in men than in women. Although the relationship reported by other authors is similar (2:1), we calculated a 3:1 ratio. The frequency of comorbidities in our population was 17%. Melo-Guzmán [23] also found a predominance in male patients (51.7%) like our own (57%).
When comparing with the results found by Halim et al. [11], 14 cases were found in association with aneurysms at different levels (25.9%), like a quarter-third (28-30%) of the population studied, however it was greater than the results found by Larios et al. (12-14%). Dural flow was observed in cases related to the parieto-occipital and temporal regions, as was observed in Koo et al. [17] and Soderman et al. [31].
García-Muñoz et al. [9] found that the most common site for AVMs was the parietal lobe (13 cases), differing from our finding of frontal lobe predominance (16 cases), followed by the parietal lobe, and an almost equal distribution for the rest of the brain (Table 2, Fig. 2). Rodríguez-Parra et al. [29] found that the most common type of the Spetzler-Martin classification was type III, which was our same conclusion.
A predilection for glues, such as cyanoacrylate (NBCA), and non-cohesive embolizing liquid, such as Onyx for the embolization of AVMs, was found, as well as an increase in recent years of the transvenous vs. transarterial approach.
Our results showed a cure rate of 33%. However, we must consider that despite having 19 success cases, five patients underwent microsurgical resection, while another seven underwent radio-neurosurgery, with a true success rate using endovascular therapy of 12.9%, a low number compared to global series. However, we must mention that the mortality reported for this analysis was 10% (approximately).
The multidisciplinary treatment of AVMs is made up of three pillar techniques: microsurgical resection, embolization, and radio-neurosurgery. It is extremely important to make an appropriate decision with an emphasis on achieving better functional outcomes for the patient. Although neurological endovascular therapy was initially used as an adjuvant treatment for neurosurgery and radio-neurosurgery, it has been used more and more frequently as the first line of treatment. There are factors that can lead to a higher rate of occlusion, such as Spetzler-Martin II, Buffalo <2, <30 years of age, as well as the use of NBCA and Onyx. Morbidity and mortality in these patients can be high, especially if there is intracranial hemorrhage, and can result in complications such as significant neurological deficit, headache, or epilepsy. Emphasis should be placed on timely detection as well as consensus for its management.
Limitations and strengths
The results should be interpreted with caution due to the retrospective nature of the data and the small sample size of this study. Although there are variations when compared to other studies such as: Saatci et al. [30] (350 cases) and Beharvahdat et al. [1] (408 cases), this article contributes to the few studies about arteriovenous malformations treated by endovascular therapy in Mexico. On the other hand, we must emphasize that, unlike the previously mentioned authors, this study was retrospective over six years, which is half of the years studied by other authors. It is necessary to continue creating databases regarding arteriovenous malformations in Mexican patients.
Much remains to be done for this type of patient, as well as the use of new technologies such as microcatheters and embolizing agents, among others, to increase the possibility of cure. It is also extremely important to understand the anatomy of these injuries, such as their afferents, efferents, and associated vascular abnormalities.
CONCLUSIONS
Intracranial AVMs are complex and rare vascular lesions with a low prevalence and incidence that can vary according to their population. It is considered important to study the behavior of this pathology in our Mexican population since there is no information in the international medical literature about age predilection, location, most frequent presentation, as well as other associated vascular alterations. Furthermore, it is of great interest to the neurological endovascular therapy sector to know the characteristics that are related to a better prognosis in embolization, whether trans arterial or transvenous, as well as the use of different embolizing materials that have emerged in recent years, not only those classically known as Onyx and NBCA, but also Squid and DMSO, among others. This work emphasizes the need for endovascular management not only as an adjunct treatment but also definitively for a better prognosis of the patient.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.