Circulation remodeling after flow diversion of an anterior communicating artery aneurysm: A case report
Article information
Abstract
Anterior communicating artery aneurysms are the most common intracranial aneurysm and have a high risk of rupture which can lead to morbidity and mortality. Traditionally, intracranial aneurysms were treated by clipping with neurosurgical access. However, certain patients may prefer less invasive approaches or not represent open surgical candidates. Flow diverters, including flow-redirection endoluminal devices (FRED), are new-generation stents that are placed endovascularly by transfemoral or transradial access. Recent studies have demonstrated that FRED is both safe and effective, with complete occlusion of aneurysms in over 90% of patients. This case highlights an interesting phenomenon of post-flow diversion circulatory remodeling, where flow diverter treatment can alter the circle of Willis anatomy and physiology.
INTRODUCTION
The anterior communicating artery (ACOM) complex is the most common location of intracranial aneurysms. Furthermore, aneurysms in this location account for the most common cause of aneurysmal subarachnoid hemorrhage and have the most complex anatomy of the anterior circulation [1,5]. Indeed, they have a higher risk for rupture than any other aneurysm within the anterior cerebral circulation according to a recent meta-analysis [10].
The formation of intracranial aneurysms is strongly associated with deficiencies in arterial walls at regions of higher wall shear stress. Wall shear stress is believed to account for the weakening of vessel walls and the formation of aneurysms [12]. The high risk of rupture of ACOM aneurysms, regardless of size, suggests the apparent need for intervention. The incidence of ACOM aneurysms has increased in recent years likely due to the improvement of minimally invasive imaging techniques [10].
The ACOM connects the bilateral anterior cerebral arteries between segments A1 and A2 [1]. Due to the proximity of the ACOM to neurological structures, anatomical variation, and flow dynamics, open neurosurgery for treatment poses both technical challenges and risks of neurological deficits [5]. There are also challenges and risks with endovascular approaches, but growing data support this treatment [1,6]. One promising strategy is endoluminal reconstruction with a flow-diverting stent. This approach offers promise for challenging anatomical locations difficult to treat by other strategies. Flow diverters are low-porosity metal stents that decrease the velocity and pressure of intra-aneurysmal blood flow. This results in delayed aneurysm thrombosis with the reconstruction of the parent artery. A flow-redirection endoluminal device (FRED), registered trademark, Microvention Inc, is a flow diverter with a low risk of complications, such as hemorrhage and thromboembolism [9]. FRED consists of an outer expanding meshwork layer and an inner layer that acts as the flow diverter [7,9]. Flow diverters, including FRED, have significantly increased the interventional treatment options for patients with intracranial aneurysms [7]; they have a high success rate for both short and long-term occlusion, up to 95.9% in some studies [7,9].
The fine mesh of FRED reduces the flow within the aneurysm body leading to clot formation. FRED also acts as a scaffold for a new intimal layer enhancing endothelialization which walls off the thrombosed aneurysm. Although the use of detachable coils has significantly increased since their approval in the 1990s, very large or wide-necked aneurysms are significantly challenging in achieving sustained occlusion with coiling alone. Flow diverters reduce the risk of aneurysm rupture by redirecting the blood flow away from the aneurysm sac, decreasing the pressure within the aneurysm [2].
CASE DESCRIPTION
A 68-year-old male with a medical history of diabetes mellitus, atrial fibrillation, hyperlipidemia, and tonsillar cancer status post resection presented with an incidental ACOM aneurysm. The ACOM aneurysm was best visualized on a left internal carotid artery angiogram, although the aneurysm was still visualized on a right internal carotid artery angiogram (see Fig. 1). As seen in Fig. 1, the ACOM aneurysm was boxed-shaped, measuring 3.9 mm wide left-to-right, 4.0 mm wide anterior-to-posterior, and 1.9 mm high neck-to-dome. The ACOM complex appeared to take on an “X” confirmation instead of an “H” shape. Of the two sides, the left anterior cerebral artery was slightly dominant.
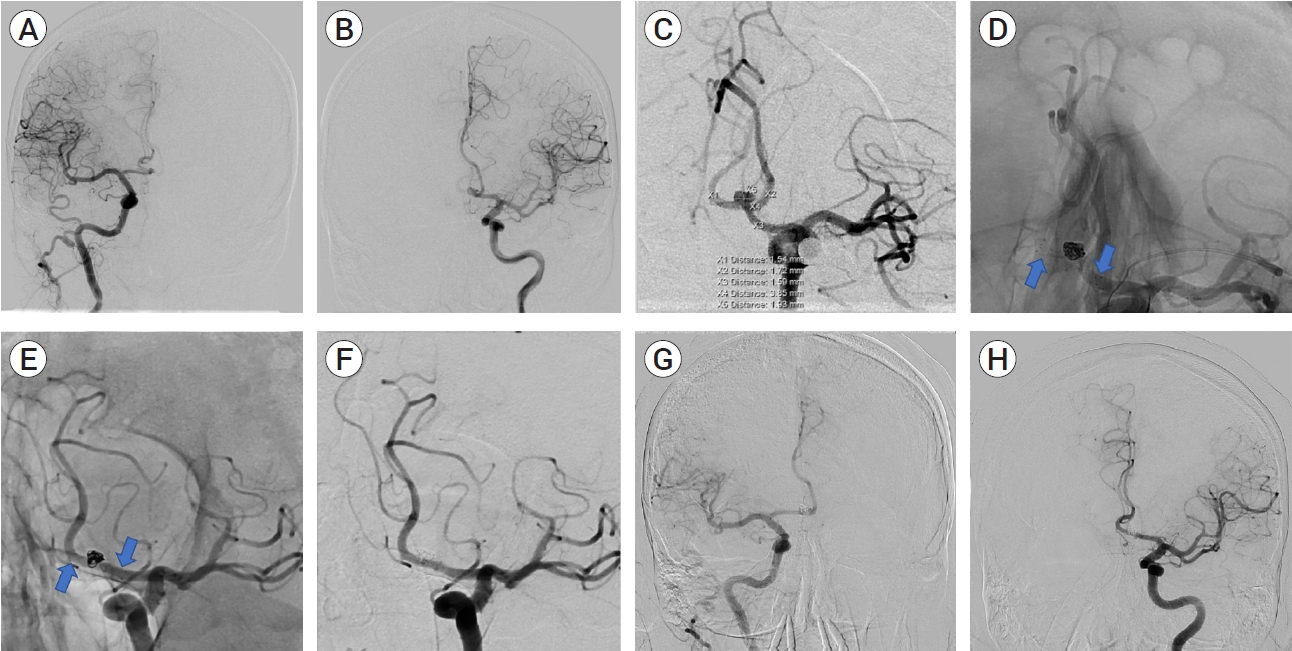
(A) Anteroposterior projection of right internal cerebral artery (ICA) contrast injection. The right anterior communicating artery (ACOM) fills from the ipsilateral right ICA. (B) Anteroposterior projection of left ICA contrast injection. The left ACOM fills from the ipsilateral left ICA. (C) Oblique working projection of left ICA contrast injection. There is a 3.9×1.9 mm ACOM aneurysm. The ACOM has an “X” configuration. (D) As the arrows indicate, native working projection immediately following treatment. There is a FRED junior stent (arrows) from the left A1 segment, covering the neck of the ACOM aneurysm, and into the right A2 segment. (E) Fifteen-month follow-up native oblique working projection. There is a FRED junior stent from the left A1 segment, covering the neck of the ACOM aneurysm, and into the right A2 segment as indicated by the arrows. (F) Fifteen-month follow-up oblique working projection of left ICA contrast injection. There is no residual aneurysm filling. (G) Fifteen-month follow-up anteroposterior projection of right ICA contrast injection. The contralateral left ACOM fills from the right ICA. (H) Fifteen-month follow-up anteroposterior projection of left ICA contrast injection. The contralateral right ACOM fills from the left ICA. FRED, flow-redirection endoluminal device
Treatment
After a multi-disciplinary discussion with the patient, a decision was made to treat the aneurysm with flow diversion. Three months after the diagnosis, the patient underwent a FRED-coil embolization. Given the aneurysm anatomy and ACOM configuration, a decision was made to place the FRED from the left A1 to the contralateral right A2 to result in the most aneurysm neck coverage. The diameter of the proximal left A1 was 2.5 mm. The distal contralateral right A2 was 2.5 mm in diameter.
Prior to the procedure, the patient received antiplatelet therapy [8]. Under general anesthesia, a sheath was placed in the common femoral artery. The left internal carotid artery (ICA) was catheterized, and a guide catheter was advanced into the cervical segment. An intermediate catheter was then advanced into the distal ICA, over a microcatheter system. Using roadmap guidance, the microcatheter over the microwire was carefully advanced through the left A1 across the ACOM complex and into the right A2. A second microcatheter was then carefully advanced into the aneurysm for coil delivery, and the first loop of the coil was placed. The FRED was then deployed across the aneurysm neck. The first coil was delivered, and a second coil was added.
Angiography revealed that the aneurysm had minimal residual filling at the base. The FRED junior flow diverting stent, had good wall apposition with good flow through the stent (see Fig. 1). The two coils were appropriately placed without any protrusion into the parent artery.
Outcome
After the FRED-coil embolization, the patient was neurologically intact and discharged in good health. The patient underwent follow-up diagnostic angiography eight and fifteen months after the embolization. At eight months post embolization, angiography revealed no evidence of residual filling of the ACOM aneurysm. The angiogram also revealed that the right A2 segment filled through the left A1 segment while the left A2 segment filled through the right A1 segment, consistent with the post-flow diversion circulatory remodeling (see Fig. 1). At this time, the patient was transitioned to monotherapy with Aspirin 81 mg. At fifteen months post embolization, angiography again revealed no evidence of residual filling related to the ACOM aneurysm. As with the last diagnostic study, the right A2 segment filled through the left A1, and the left A2 segment filled through the right A1 (see Fig. 1).
DISCUSSION
ACOM aneurysms are complex and difficult to treat. Due to their high risk of rupture which can lead to neurological deficits, intervention is often recommended. Traditional open neurosurgical interventions, such as clipping, remain an option but pose some challenges. Endovascular embolization offers another approach, and there is a growing toolset available to aid in the treatment of complicated aneurysms. Indeed, the outcomes after endovascular embolization, such as the use of a FRED, have a similar success rate to open surgery [1,5,6,10,12]. Flow diverters have increasingly become a treatment of choice due to high cure rates and low rates of complications [3]. FRED is one option that provides stable positioning and good aneurysm coverage when major arterial branches are located near the aneurysm [8]. Flow diverters cover the intersection of the parent artery and the aneurysm, decreasing the entry of blood into the aneurysm through a low-permeable dual-layer stent [3]. The blood flow will be restricted from entering the aneurysm once the perpendicular force of the FRED is greater than the hemodynamic force. Another mechanism of FRED is the promotion of endothelialization in the parent artery to isolate artery from aneurysm. Flow diverter design plays a critical role in successful endothelialization [2].
This case further highlights the interesting and variable phenomenon of post-flow diversion circulatory remodeling [4,8]. Arterial remodeling represents both physiologic and pathologic changes in the vessel wall structure, usually in response to a change in pressure [11]. Although remodeling is seen in the creation of an aneurysm, FRED facilitates arterial remodeling in the treatment of the aneurysm. Intervention to remodel the arterial wall will result in occlusion of the aneurysm with little to no risk of recurrence if endothelial coverage is complete [8]. Arterial remodeling occurs as a result of said endothelialization, reduced hemodynamic forces within the aneurysm, and flow diversion due to the device. A flow diverter induces a beneficial physiological response to remodel the parent vessel structure. The low flow state within the aneurysm results in coagulation, leading to thrombosis within the aneurysm [11]. Knowing that remodeling is simply a homeostatic mechanism to adapt to changes exerted on the vessel wall, flow diverters can be used to exploit this process.
EuFRED, the largest study to date evaluating FRED, reported a complete occlusion rate in 91.3% of cases after 1 year [3]. Mortality with FRED was reported to be less than 1% and morbidity was reported at 1.5%. EuFRED and other smaller studies found FRED to be safe and effective for major vessels of the circle of Willis [3]. This case also highlights the role flow diverters, and perhaps specifically the dual-layered FRED, have in circulation remodeling. The ACOM was remodeled such that the right A1 supplied the left A2 and the left A1 supplied the right A2. Other data support that “jailing” vessels of substantial size or those which subtend critical tissue remain patent by demand [11].
CONCLUSIONS
This case report highlights the successful use of flow diversion for the treatment of a complex aneurysm of the ACOM complex. Flow diverters, such as FRED, are becoming a widely used approach for the treatment of aneurysms which are less suited to conventional open or endovascular techniques. Importantly, this case exemplifies the robust and variable phenomenon of post-flow diversion circulatory remodeling.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
