 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 23(3); 2021 > Article |
|
Abstract
Objective
The aim of this nationwide age- and sex- matched longitudinal follow up study is to determine the risk of ParkinsonŌĆÖs disease (PD) associated with ischemic stroke in Korea.
Methods
Patient data were collected from the National Health Insurance Service-National Health Screening Cohort (NHIS-HEALS). PD was identified using the International Classification of Diseases (ICD) 10-CM code G 20. In total, 6,475 patients were enrolled in the PD group from the NHISS. After subtracting 1,039 patients who underwent hospitalization less than once or those who visited an outpatient clinic less than two times, 5,259 patients who were diagnosed after January 1, 2004 ultimately participated in this study. After case-control match was done through 1:5 age- and sex- stratified matching, 26,295 individuals were chosen as control. Kaplan-Meier method and Cox proportional hazard regression analysis were performed to evaluate the risk of ischemic stroke in PD.
Results
The hazard ratio of ischemic stroke in the PD group was 3.848 (95% confidence interval (confidence interval [CI]): 3.14-4.70) after adjusting for age and sex. The adjusted hazard ratio of ischemic stroke in PD group was 3.885 (95% CI: 3.17-4.75) after adjusting for comorbidities. According to subgroup analysis, in male and female and non-diabetes and diabetes and non-hypertension and hypertension and dyslipidemia and non-dyslipidemia subgroups, ischemic stroke incidence rates were significantly higher in the PD group than those in the control group.
ParkinsonŌĆÖs disease (PD) is one of the most common progressive neurodegenerative disease in the elderly people [4]. In previous studies, it was found that ischemic heart disease accounts for the highest mortality rate in patients with ParkinsonŌĆÖs disease [21,22]. However, the risk of ischemic stroke in PD patients remains unclear. Some studies have found that PD is associated with an increased risk of ischemic stroke and an increase in ischemic stroke-related mortality [2,3,6,11]. In contrast, other studies have found that PD patients have lower risk of ischemic stroke [15,23,27].
Most studies about the relationship between ParkinsonŌĆÖs disease and ischemic stroke were small size sample based longitudinal cohort studies [2,11,15,27]. Except for two studies [11,26], the rest ones were studied in the western countries. Among them, the study conducted in Korea was limited to a small size cross sectional study [26].
This nationwide longitudinal cohort study aimed to investigate the risk of ischemic stroke in PD in Korea.
The Republic of Korea has a single-payer health insurance system, managed by the National Health Insurance Sharing Service (NHISS) [1,14,16,19]. All health-care providers must submit medical claims to the NHISS for review and reimbursement. The NHISS also provides national health examinations for those who are aged Ōēź40 years, biannually for office workers and annually for non-office workers. Therefore NHISS can provide database representing a cohort who participates in national health examinations [24]. The NHISS claim database includes extensive information about demographics, medical treatments, medical procedures, and various disease diagnoses according to the 10th revised codes of the International Classification of Diseases (ICD-10) [13]. Researchers who are approved by the official review committee can use this NHISS database, and we acquired the rights to use it from the institutional review board of the CHA Bundang Medical Center of CHA University (IRB No. 2020-01-011).
Our analysis of age- and sex- matched cohorts was designed to estimate the risk of ischemic stroke in PD patients. The study population included a PD group and a control group. The Korea NIHSS cohort included 515,547 participants who followed up for 12 years, until December 2015. We extracted medical claims and demographic information from the NHISS database, including age, sex, household income level, and disease codes using the ICD-10 codes. We evaluated the risk of ischemic stroke after adjusting for age, sex, and comorbidities specifically hypertension, diabetes mellitus, and dyslipidemia. Information on these preexisting comorbid medical disorders was obtained by reviewing all of the outpatient and inpatient records from the NHISS database [20].
We extracted 6,475 PD subjects from a total of 515,547 patients in the NHISS Database. The occurrence of PD was defined by the code G20 based on the ICD-10 code. To include patients who had more disease activity, 5,436 people who were hospitalized more than 1 time or who visited outpatient clinic more than 2 times were selected. After removing 177 subjects with preexisting PD, a total of 5,259 patients with newly diagnosed after 2004.01.01 remained. Through 1:5 age- and sex-stratified matching without replacement, using a greedy match algorithm of the R package ŌĆśMatch ITŌĆÖ, 26,295 individuals were chosen as controls [8,9]. Both groups were followed up until December 31, 2015 (Fig. 1).
The Chi-square test and StudentŌĆÖs t-test were used to compare mean differences in the demographic characteristics and comorbidities between the PD and control groups (Table 1). The ischemic stroke-free survival probabilities of the two groups were estimated using the Kaplan-Meier method. Differences in disease-free survival between the two groups were tested using the WilcoxonŌĆÖs log rank test. Multivariate analyses with a Cox proportional-hazards regression model were conducted to estimate the effect of PD on the subsequent occurrence of each event. We used two Cox proportional-hazards regression models: In model 1; age and sex were adjusted. In model 2; age, sex, low income and other comorbidities were adjusted (Table 2). In each subgroup, the Cox-proportional-hazard regression model was used to estimate the effect of PD on the subsequent occurrence of each event (Table 3). The analyses were performed using R software (version 3.3.3, R foundation for statistical computing, Vienna, Austria).
We identified a total of 5,259 individuals with newly diagnosed PD. The mean age was 62.87┬▒8.42 years, and majority of the patients were male (53.28%). There were significant differences between the two groups in terms of the prevalence of low income (p=0.001), diabetes mellitus (p<0.001), and hypertension (p<0.001) (Table 1).
The incidence rate of ischemic stroke was significantly higher in the PD group than in the control group (p<0.001, Fig. 2). The Kaplan-Meier curves with cumulative hazards of ischemic stroke showed that the PD group had a higher risk of developing ischemic stroke than the control group. In a multivariate analysis of Cox proportional-hazards regression model 1, the hazard ratio of ischemic stroke in the PD group was 3.848 compared with that in the control group (95% confidence interval [CI]: 2.64-5.68, Table 2). In the multivariate analyses of model 2, the hazard ratio of ischemic stroke in the PD group was 3.885 (95% CI: 2.86-6.19, Table 2).
In both male and female subgroups, ischemic stroke incidence rate showed difference between the PD and control group (95% CI: 2.814-5.008 and 2.975-5.214, respectively, Table 3). In both age subgroups (<65 and Ōēź65), ischemic stroke incidence rates were significantly different between the PD and the control group (95% CI, 4.152-8.330 and 2.420-3.986, respectively, Table 3). In both the non-diabetes subgroup and diabetes subgroup, the ischemic stroke incidence rate was significantly different between the PD and control group (95% CI, 2.833-4.492, and 3.058-6.997, respectively, Table 3). In both the non-hypertension and hypertension subgroups, the ischemic stroke incidence rate was significantly different between the PD and control group (95% CI, 2.969-5.571 and 2.881-4.856 respectively, Table 3). In both the non-dyslipidemia and dyslipidemia subgroups, the ischemic stroke incidence rate was also significantly different between the PD and the control group (95% CI, 3.141-4.933 and 2.231-5.406 respectively, Table 3).
This study was a nationwide longitudinal cohort study based on the NHISS database. In this nationwide cohort study, 5,259 PD patients had a significantly higher risk of ischemic stroke. The risk of ischemic stroke is similar to what has been reported in previous studies [2,3,6,11,26]. One population based follow up study in Taiwan reported that PD group had a 2.10 times higher of ischemic stroke incidence rate than control group [11]. Another cross sectional study in Korea showed that PD patients without other ischemic stroke risk factors had a significantly higher risk of ischemic cerebrovascular diseases compared to healthy controls [1,26]. In this study, the incidence rate of ischemic stroke was found to be significantly increased in PD patients, similar to previous studies.
The mechanism of increased ischemic stroke in PD patients is unclear. One of the reasonable theories is that oxidative stress is present in idiopathic PD, and these oxidative damage products interfere with cellular function [7,12]. Oxidative stress is one of explainable pathogenesis, as it can damage dopaminergic cells of substantia nigra. Oxidative stress accumulated into endothelial cell cause atherosclerotic change, which may increase the risk of cerebrovascular accidents. Thus, existence of PD may indicate higher oxidative stress accumulation, which bring about higher incidence rate of ischemic stroke. Second, Orthostatic hypotension occurred by preexisting PD may lead to ischemic brain parenchymal damage [18,28]. Orthostatic hypotension can result from autonomic dysfunction in PD. One meta-analysis reported that the prevalence of orthostatic hypotension was 30% of patients with PD [28]. Another cohort study showed 48% of PD patient diagnosed for 20 years had orthostatic hypotension [17].
Several limitations of this study should be presented. First, chronic infectious burden and elevated inflammatory markers, such as C-reactive protein and interleukin-6, can be associated with the development of atherosclerosis and increased risk of ischemic stroke [5,10,25]. However, data regarding inflammatory markers lack in the National Health Insurance database. Therefore, it is difficult to evaluate the potential effects of inflammatory markers on the association between PD and ischemic stroke. Second, variables in the healthcare claim data cannot accurately reflect patientŌĆÖs medical conditions [5,10,24,25].
Even with this limitation, this is the first nationwide longitudinal cohort study to evaluate the relevance of PD with ischemic stroke in Korea.
ACKNOWLEDGEMENTS
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2020R1I1A1A01072258). This work was also supported by a grant of Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and future Planning (NRF-2020R1F1A1069875).
Fig.┬Ā1.
Flow diagram of the cohort creation process. This study was a 12-year longitudinal cohort study established with the NHISS cohort. NHISS, National Health Insurance Sharing Service; PD, ParkinsonŌĆÖs disease; ICD, International Classification of Diseases.
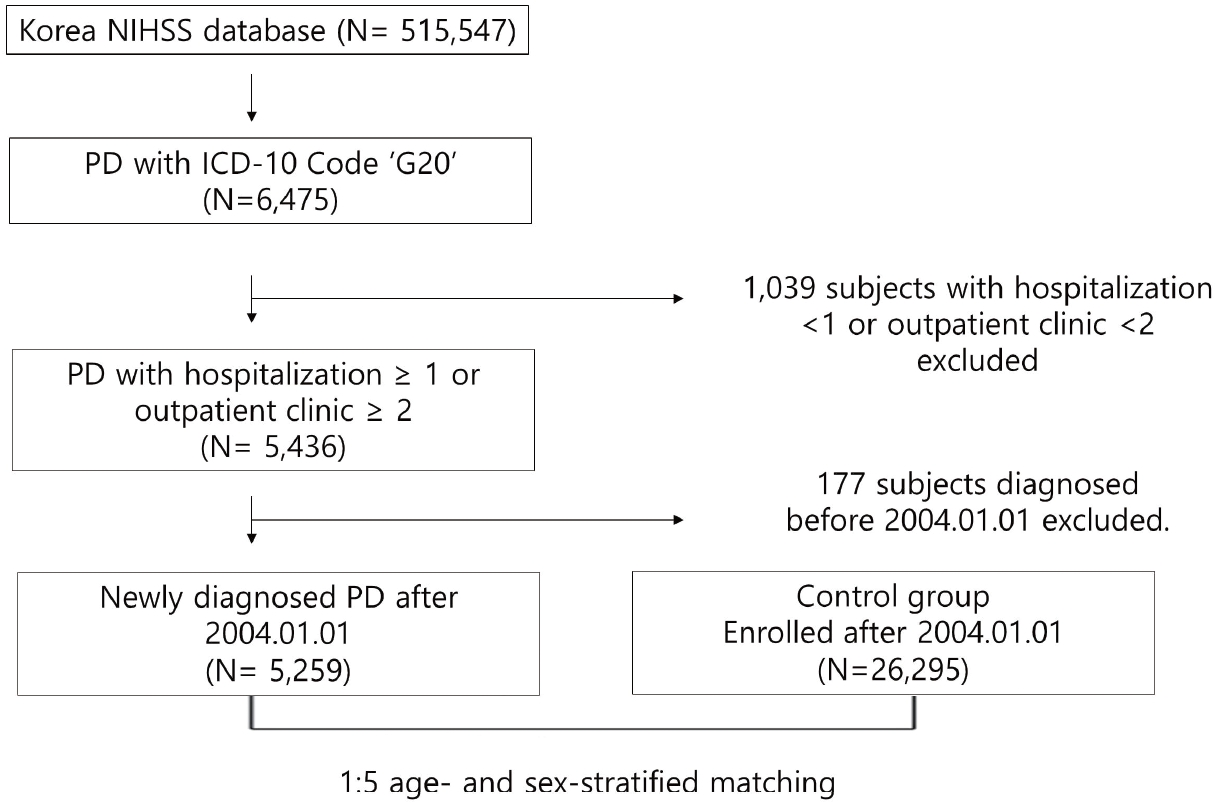
Fig.┬Ā2.
Comparison of the cumulative incidence rate of ischemic stroke in the PD and control group. The Kaplan-Meier curves with cumulative hazards of ischemic stroke were compared between PD and control group. PD, ParkinsonŌĆÖs disease.

Table┬Ā1.
Characteristics in the PD and control group
Table┬Ā2.
Adjusted hazard ratio for ischemic stroke event in the PD and control group
Table┬Ā3.
Ischemic stroke incidence in subgroup analyses between PD and control group
REFERENCES
1. Bae KH, Hong JB, Choi YJ, Jung JH, Han IB, Choi JM, et al. Association of congestive heart failure and death with ankylosing spondylitis: a nationwide longitudinal cohort study in Korea. J Korean Neurosurg Soc. 2019 Mar;62(2):217-24.



2. Becker C, Jick SS, Meier CR. Risk of stroke in patients with idiopathic Parkinson disease. Parkinsonism Relat Disord. 2010 Jan;16(1):31-5.


3. Ben-Shlomo Y, Marmot MG. Survival and cause of death in a cohort of patients with parkinsonism: possible clues to aetiology? J Neurol Neurosurg Psychiatry. 1995 Mar;58(3):293-9.



4. de Lau LM, Breteler MM. Epidemiology of ParkinsonŌĆÖs disease. Lancet Neurol. 2006 Jun;5(6):525-35.


5. Elkind MS, Ramakrishnan P, Moon YP, Boden-Albala B, Liu KM, Spitalnik SL, et al. Infectious burden and risk of stroke: the northern Manhattan study. Arch Neurol. 2010 Jan;67(1):33-8.


6. Gorell JM, Johnson CC, Rybicki BA. ParkinsonŌĆÖs disease and its comorbid disorders: an analysis of Michigan mortality data, 1970 to 1990. Neurology. 1994 Oct;44(10):1865-8.


7. Harrison D, Griendling KK, Landmesser U, Hornig B, Drexler H. Role of oxidative stress in atherosclerosis. Am J Cardiol. 2003 Feb;91(3A):7A-11A.


8. Ho D, Imai K, King G, Stuart EA. MatchIt: nonparametric preprocessing for parametric causal inference. J Stat Soft. 2011 May;42(8):1-28.

9. Ho DE, Imai K, King G, Stuart EA. Matching as nonparametric preprocessing for reducing model dependence in parametric causal inference. Political Analysis. 2007 15(3):199-236.

10. Hoshi T, Kitagawa K, Yamagami H, Furukado S, Hougaku H, Hori M. Relations of serum high-sensitivity C-reactive protein and interleukin-6 levels with silent brain infarction. Stroke. 2005 Apr;36(4):768-72.


11. Huang YP, Chen LS, Yen MF, Fann CY, Chiu YH, Chen HH, et al. ParkinsonŌĆÖs disease is related to an increased risk of ischemic stroke-a population-based propensity score-matched follow-up study. PLoS One. 2013 Sep;8(9):e68314.



13. Kang SH, Choi EK, Han KD, Lee SR, Lim WH, Cha MJ, et al. Underweight is a risk factor for atrial fibrillation: A nationwide population-based study. Int J Cardiol. 2016 Jul;215:449-56.


14. Kim YS, Kim JG, Yi J, Choi JM, Chung CK, Choi UY, et al. Changes in the medical burden of pyogenic and tuberculous spondylitis between 2007 and 2016: A nationwide cohort study. J Clin Neurosci. 2020 Mar;73:89-93.


15. Korten A, Lodder J, Vreeling F, Boreas A, van Raak L, Kessels F. Stroke and idiopathic ParkinsonŌĆÖs disease: does a shortage of dopamine offer protection against stroke? Mov Disord. 2001 Jan;16(1):119-23.


16. Lee DH, Choi YJ, Han IB, Hong JB, Do Han K, Choi JM, et al. Association of ischemic stroke with ankylosing spondylitis: a nationwide longitudinal cohort study. Acta Neurochir (Wien). 2018 May;160(5):949-55.


17. Lim SY, Lang AE. The nonmotor symptoms of ParkinsonŌĆÖs disease--an overview. Mov Disord. 2010 25 Suppl 1:S123-30.


18. McDonald C, Newton JL, Burn DJ. Orthostatic hypotension and cognitive impairment in ParkinsonŌĆÖs disease: Causation or association? Mov Disord. 2016 Jul;31(7):937-46.


19. Park CJ, Choi YJ, Kim JG, Han IB, Do Han K, Choi JM, et al. Association of acute myocardial infarction with ankylosing spondylitis: a nationwide longitudinal cohort study. J Clin Neurosci. 2018 Oct;56:34-7.


20. Park CS, Choi EK, Han KD, Lee HJ, Rhee TM, Lee SR, et al. Association between adult height, myocardial infarction, heart failure, stroke and death: a Korean nationwide population-based study. Int J Epidemiol. 2018 Feb;47(1):289-98.


21. Rektor I, Goldemund D, Sheardov├Ī K, Rektorov├Ī I, Mich├Īlkov├Ī Z, Dufek M. Vascular pathology in patients with idiopathic ParkinsonŌĆÖs disease. Parkinsonism Relat Disord. 2009 Jan;15(1):24-9.


22. Roos RA, Jongen JC, van der Velde EA. Clinical course of patients with idiopathic ParkinsonŌĆÖs disease. Mov Disord. 1996 May;11(3):236-42.


23. Scigliano G, Musicco M, Soliveri P, Piccolo I, Ronchetti G, Girotti F. Reduced risk factors for vascular disorders in Parkinson disease patients: a case-control study. Stroke. 2006 May;37(5):1184-8.


24. Seong SC, Kim YY, Park SK, Khang YH, Kim HC, Park JH, et al. Cohort profile: the National Health Insurance Service-National Health Screening Cohort (NHIS-HEALS) in Korea. BMJ Open. 2017 Sep;7(9):e016640.



25. Simanek AM, Dowd JB, Pawelec G, Melzer D, Dutta A, Aiello AE. Seropositivity to cytomegalovirus, inflammation, all-cause and cardiovascular disease-related mortality in the United States. PLoS One. 2011 Feb;6(2):e16103.



26. Song IU, Lee JE, Kwon DY, Park JH, Ma HI. ParkinsonŌĆÖs disease might increase the risk of cerebral ischemic lesions. Int J Med Sci. 2017 Mar;14(4):319-22.



- TOOLS
-
METRICS

-
- 5 Crossref
- 0 Scopus
- 3,191 View
- 104 Download
- ORCID iDs
-
Seil Sohn

https://orcid.org/0000-0001-5724-8099 - Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



