Clinical analysis of young adult patients with ruptured intracranial aneurysms: a single-center study of 113 consecutive patients
Article information
Abstract
Objective
Intracranial aneurysm most frequently occurs in old aged patients; however, aneurysmal subarachnoid hemorrhage (SAH) has been occasionally reported in young adults. This study aimed to compare patients with intracranial aneurysms younger than 40 years with those ≥40 years and investigates the characteristics of their ruptured aneurysm.
Methods
We retrospectively collected the data of 389 patients (103 patients aged 20 to 39 years; 286 patients aged above 40 years) who were treated for ruptured cerebral aneurysms between January 2008 and December 2018 at our institution.
Results
The young adult patients were more often men and were smokers as compared to controls (63.1% vs. 39.2%, 40.5% vs. 36.0%, respectively). The young adult patients showed a tendency for lower Hunt and Hess grade, modified Fisher grade, less postoperative morbidity, and better clinical outcome. Aneurysm ruptures occurred most frequently in the anterior cerebral artery in both groups, but aneurysms ruptures in the internal carotid artery and posterior circulation was infrequent among young adult patients. The aneurysms in young adult patients were more often elongated or irregularly shaped (82.5% vs. 64.0%, respectively) and were significantly smaller (5.9 mm) than that in controls (6.8 mm).
Conclusions
Ruptured aneurysms of young adult patients have unique characteristics with regard to its preponderance in male smoker, elongated or irregular shape and relatively smaller size.
INTRODUCTION
Intracranial aneurysms most frequently occur in old aged patients.12) This tendency suggests that hemodynamic stress in the intracranial vasculatures is a causative factor of the intracranial aneurysm formation and rupture in adults. However, rupture of aneurysms has been occasionally reported in patients younger than 40 years. The incidence of aneurysm rupture in patients younger than 40 years is 10–20% of all patients with ruptured aneurysm.2)12)19) Studies have reported the characteristics of ruptured aneurysms in the general population; however, studies on young adult patients are limited.8)21)27) We hypothesized that ruptured aneurysms in patients younger than 40 years may have unique characteristics associated with rupture, unlike those in patients aged more than 40 years, such as atherosclerosis, hypertension, etc. To investigate the nature of these features, we aimed to compare between the characteristics of ruptured intracranial aneurysms in patients younger than 40 years and those in patients ≥40 years.
MATERIALS AND METHODS
We collected the data retrospectively from the medical records of 956 patients treated for ruptured intracranial aneurysms between January 2008 and December 2018 at our institution. Among them, 113 (11.8%) patients aged 20 to 39 years were selected as cases and 307 patients aged above 40 years were randomly selected as controls. Seven patients with blister aneurysm and 24 patients with ruptured dissection were excluded because of difficulty in measuring the size of the aneurysm; eventually, 389 patients with saccular aneurysms were included for further analyses. The study cohort consisted of hospitalized patients, and thus, cases of sudden death due to subarachnoid hemorrhage (SAH) were excluded from the analyses. The study was conducted with the approval of our institutional review board (AJIRB-MED-MDB-18-433), and informed consent was obtained from each patient or their family.
The following data were collected for each patient: age, sex, body mass index (BMI), social history (smoking), medical history (hypertension/coronary artery occlusive disease/heart failure, diabetes mellitus, renal failure, and systemic lupus erythematosus), familial history of cerebrovascular disease, initial systolic blood pressure, Hunt and Hess grade (HHG), modified Fisher grade, type of therapy, and radiologic characteristics of aneurysms (location, multiplicity, morphology, and size). The morphology of intracranial aneurysms had been evaluated using a multi-detector computed tomography angiography (Brilliance; Philips Healthcare, Amsterdam, Netherlands) and three-dimensional digital subtraction angiography (Allura Xper FD20/10; Philips Healthcare, Amsterdam, Netherlands). The aneurysm size was defined as the broadest dimension on three-dimensional view.
The patients underwent endovascular procedure or aneurysm clipping. The endovascular procedures were performed under general anesthesia.
The clinical outcome for each patient was evaluated by measuring Glasgow Outcome Scale (GOS) score at discharge.
Statistical analyses
All statistical analyses were performed using IBM SPSS Statistics version 19 (IBM Corp., Armonk, NY, USA). Categorical variables are expressed as proportions, and continuous variables are expressed as means and standard deviations. Age differences were assessed using the Student’s t test for parametric variables, and using the Wilcoxon rank-sum test for nonparametric variables. The study groups were analyzed using the χ2 test or Fisher’s exact test for cross-tabulation. The results of all statistical tests are reported with a 5% level of significance.
RESULTS
Clinical characteristics
Table 1 shows the clinical characteristics of patients with ruptured intracranial aneurysms.
One hundred and three (26.5%) patients were aged 20 to 39 years. The median age was 50 (range, 22–89) years and 177 (45.5%) patients were men. The mean BMI was 23.6 (13.3–35.9) kg/m2, and 153 (39.3%) of the patients were smokers.
One hundred and eight (27.8%) patients had SAH of HHG IV–V and 130 (33.4%) had SAH of modified Fischer grade IV.
Among all cases examined, 188 (48.3%) underwent endovascular coiling and 201 (51.7%) underwent surgical clipping.
One hundred and twenty-three (31.6%) patients showed poor clinical outcomes on the GOS with scores below 3.
Ninety-seven (24.9%) patients experienced postoperative morbidity; hydrocephalus that required surgical treatment (n=51), severe brain swelling or vasospasm-induced infarction that required surgical decompression (n=25), central nervous system infections (n=1), intraprocedural thrombosis/unintended vessel occlusion (n=2), rebleeding (n=1), and other medical problems (e.g., pneumonia, sepsis, acute kidney injury) (n=17).
Radiologic characteristics
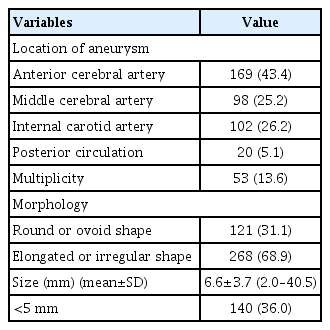
Table 2 shows the radiologic characteristics of patients with ruptured intracranial aneurysms.
The most commonly affected artery was the anterior cerebral artery (ACA) (n=169; 43.4%), followed by the internal carotid artery (ICA) (n=102; 26.2%), middle cerebral artery (MCA) (n=98; 25.2%), and posterior circulation (n=20; 5.1%). ACA aneurysms included aneurysms originating in the proximal ACA (n=4), anterior communicating artery (n=151), and distal ACA (n=14). ICA aneurysms included aneurysms originating in the ICA bifurcation (n=5), anterior choroidal artery (n=20), posterior communicating artery (n=73) and paraclinoid ICA (n=4).
Fifty-three (13.6%) patients had at least two intracranial aneurysms.
The morphology of aneurysms showed the following distribution: Round or ovoid shape (n=121; 31.1%) and elongated or irregular shape (n=268; 68.9%).
The mean size of aneurysms was 6.6 mm in all 389 patients, and 140 (36.0%) aneurysms were smaller than 5 mm.
Clinical and radiologic characteristics in cases and controls
Table 3 shows the characteristics of patients with ruptured aneurysms according to age.
The young adult patients significantly comprised more men and smokers (p<0.001; and p=0.026, respectively); however, BMI of the patients showed no association with ruptured aneurysms. The patients’ medical history correlated significantly with age (p<0.001).
The young adult patients showed a significant tendency for lower HHG, modified Fisher grade, and higher GOS score (p<0.001, p<0.001, and p=0.004, respectively). In addition, incidence of morbidity was significantly lower in young adult patients (p=0.042), and experienced less frequent presentations of postoperative hydrocephalus and other medical complications.
Among the data of young adult patients examined, SAH secondary to the ruptured aneurysms developed in the ACA in 49 (47.6%) patients, in the MCA in 32 (31.1%) patients, in the ICA in 25 (20.4%) patients, and in the posterior circulation in 1 (1.0%) patients. In contrast, among the control group patients, SAH secondary to the ruptured aneurysms developed in the ACA in 120 (42.0%) patients, in the ICA in 81 (28.3%) patients, in the MCA in 66 (23.1%) patients, and in the posterior circulation in 19 (6.6%) patients. There was a significant difference in the location of the ruptured aneurysms. The aneurysms ruptures occurred most frequently in the ACA in both groups, but aneurysms ruptures in the ICA and posterior circulation was infrequent among young adult patients.
The aneurysms were significantly more often elongated/irregularly shaped in young adult patients (p<0.001).
The aneurysm size was also associated with age. The ruptured aneurysms in young adult patients were statistically significant smaller (mean size, 5.9 mm) than that in controls (mean size, 6.8 mm; p=0.033).
DISCUSSION
Demographic characteristics of young adult patients
Intracranial aneurysms are influenced by environmental factors. Smoking showed statistically significant associations with an increased risk of SAH.5)15) Obesity, as a widely recognized risk factor for cerebrovascular disease, may also increase the risk of SAH. In the literature,4) otherwise, low BMI is independent risk factor of SAH.
We hypothesized that smoking and being overweight/underweight may be associated with aneurysm ruptures in young adult patients. In our cases series, smoking was more frequently observed among young adult patients, but had no significant difference on adjustment of confounders. Additionally, all the patients with ruptured aneurysms were overweight, but there was no significant difference according to age.
Favorable outcomes in young adult patients
Young adult patients with ruptured aneurysm usually show good prognosis with rare cases of perioperative mortality.18)20) Clinical outcomes among young adults with ruptured aneurysms are generally favorable because of the low incidences of hydrocephalus, severe vasospasm, and other medical problems.12)13)19)
The present study showed similar findings; young adult patients had less medical problems such as hypertension or diabetes mellitus than the comparison group (20.4% vs. 44.1%, respectively). The only 20.4% of the young adult patients had poor clinical outcomes with GOS scores below 3, and the perioperative morbidity was only 17.5% compared to controls.
Male predominance in young adult patients and discrepancy of aneurysm location
Previous studies have reported a sex difference in the prevalence of intracranial aneurysm.7)8)19)27) Aneurysms in children and adolescents are more frequent in men, whereas in women, the predominance is more during adulthood.
In the present study, 63.1% of the young adult patients with ruptured aneurysms were men, outnumbering that in controls by nearly a factor of 1.5.
Female sex is an independent risk factor for the growth and rupture of aneurysm5)25) and estrogen deficiency, especially in postmenopausal women, has an important impact on the pathophysiology of the formation and rupture of aneurysms.5)24) Estrogen has been shown to influence vascular endothelial function, and enhance normal physiological vascular system.6)23) By preserving the vascular integrity, therefore, estrogen in young adult women appears to play a significant protective role in the formation and rupture of aneurysms. That is, ruptured aneurysms were more predominant among young adult men with relatively low estrogen levels than women.
Moreover, the present study shows that the location of aneurysm differed among young adult patients. The aneurysms ruptures occurred most frequently in the ACA in both groups, and in young adult patients, besides the affected artery, the MCA (31.1%) and ICA (20.4%) were prominent locations for the development of aneurysm. Contrarily, among controls, the rupture of aneurysms was more likely to occur in the ICA (28.3%) and MCA (23.1%).
This anatomic discrepancy may be explained by the predominance of male sex among the young adult patients. Previous studies1)6)13)19) reported sex differences in the anatomical distribution of ruptured aneurysms; aneurysms in the ACA were reportedly overrepresented in men, and that in the ICA were overrepresented in women.
Size and morphology of aneurysm in young adult patients
Recent studies have indicated that rupture of intracranial aneurysm is primarily dependent on the aneurysm size,9–11) and risk of rupture increases substantially with the increasing aneurysm size.17)26)
Recommendations of the American Heart Association suggest that the rupture risk of incidental aneurysms smaller than 10 mm is low.3) According to the International Study of Unruptured Intracranial Aneurysms 2 study,26) in the anterior circulation, the rupture rate of aneurysms smaller than 7 mm was 0–0.3% per year, while that of aneurysms of size 7–12 mm was 0.5% per year.
However, in the present study, 94.2% of ruptured aneurysms in young adult patients were smaller than 10 mm and 47.6% were smaller than 5 mm. In young adult patients, the size of ruptured aneurysms was significantly smaller than that in controls.
The pathophysiology of intracranial aneurysm formation and development can explain the small size of the ruptured aneurysms among young adult patients in this study. Previous studies found that the rupture risk of intracranial aneurysms was highest at the first year of diagnosis and decreased thereafter. In the initial stage of aneurysm formation, aneurysm rupture can occur because of the weak aneurysm wall. After this initial stage, healing of aneurysm wall occurs and the aneurysms may achieve some stability.14)22) Therefore, the small intracranial aneurysms of young adult patients who are first diagnosed could be require thorough assessment and close follow-up.
In general, irregular shape indicates increased risk of aneurysmal rupture. Lindgren et al.16) reported that irregular shape is strongly associated with rupture in cerebral aneurysms and independent of patient background including their age. The present study revealed a similar finding, indicating that 68.9% of total ruptured aneurysms have elongated or irregular shape of aneurysms.
Limitations
This study was limited by its retrospective nature and selection bias. To further evaluate these observations, future studies that include young adult patients with unruptured aneurysms are needed.
CONCLUSIONS
The ruptured aneurysms of young adult patients have unique characteristics that include a strong predominance for male smoker, elongated or irregular shape and small size.
Notes
Disclosure
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.