A Meta-analysis for Evaluating Efficacy of Neuroendoscopic Surgery versus Craniotomy for Supratentorial Hypertensive Intracerebral Hemorrhage
Article information
Abstract
Objective
Hypertensive intracerebral hemorrhage is a potentially life-threatening neurological deficit with the highest morbidity and mortality. In recent years, neuroendoscopy has been used to treat intracerebral hemorrhages (ICHs). However, the choice of neuroendoscopic surgery or craniotomy for patients with ICHs is controversial. The objective of this meta-analysis was to assess the efficacy of neuroendoscopic surgery compared to craniotomy in patients with supratentorial hypertensive ICH.
Materials and Methods
A systematic electronic search was performed using online electronic databases such as Pubmed, Embase, and Cochrane library updated on December 2017. The meta-analysis was performed by only including studies designed as randomized controlled trials.
Results
Three randomized controlled trials met our inclusion criteria. Pooled analysis of death showed that neuroendoscopic surgery decreased the rate of death compared to craniotomy (RR=0.58, 95% CI: 0.26–1.29; P=0.18). Pooled results of complications showed that neuroendoscopic surgery tended to have fewer complications than craniotomy had (RR=0.37, 95% CI: 0.28–0.49; P < 0.0001).
Conclusion
Although the presenting analyses suggest that neuroendoscopic surgery should have fewer complications than craniotomy dose, it had no superior advantage in morbidity rate definitely. Therefore, it may be necessary for the neurosurgeons to select best optimal patients for individual treatment.
INTRODUCTION
Hypertensive intracerebral hemorrhage (HICH) is a condition that can cause high mortality rate7) and severe disabilities22)23). Management of primary intracerebral hemorrhage remains controversial. Previous studies have shown that neurosurgical treatment rather than conservative treatment is associated with better outcomes (P < 0.001)17) for HICH. To treat HICH, surgical procedures have been employed. However, their results are not strongly established as well as heterogeneous among studies. To dates, several studies have reported different strategies of neurosurgical interventions, including craniotomy and neuroendoscopic surgery2)3)21)24).
In the past several decades, craniotomy played critical roles in treatment of HICH. However, several prospective randomized controlled trials have failed to show benefit of craniotomy for treating HICH patients16)21). Endoscopic surgery has been also applied in the treatment of HICH in recent years. Many studies have suggested that endoscopic evacuation of HICH should have better outcomes and more safety for HICH patients5)18) than traditional craniotomy23)26). However, due to retrospective nature of research or limited sample size, no concrete conclusion could be drawn about effects of endoscopic surgery on outcomes of HICH patients1)25).
Therefore, the objective of this meta-analysis was to compare craniotomy and neuroendoscopic surgery to determine which one might be safer and more effective in promoting outcomes and reducing complications in patients with HICHs.
MATERIALS AND METHODS
Retrieval strategy
Published articles that compared efficacy and safety of neuroendoscopic surgery and craniotomy in patients with supratentorial HICH up to December 2017 were retrieved. Searchable databases included Pubmed, Embase, and Cochrane library. The process was established to find all articles based on MeSH terms and keywords of “craniotomy”, “neuroendoscopic surgery” and “hypertensive intracerebral hemorrhage”. No limitation was used during literature search. We identified full-text papers from reference materials for further evaluation.
Eligibility criteria
Articles that met the following inclusion criteria were included in this analysis: (1) studies that were designed as randomized controlled trials; (2) articles that enrolled hypertensive intracerebral hemorrhage patients; (3) trials that compared craniotomy to neuroendoscopic surgery; and (4) studies that provided data of perioperative morbidity or mortality. Studies that did not meet the above inclusion criteria were excluded from meta-analysis.
Quality assessment
Two investigators independently rated the quality of retrieved studies. Risk of bias items (ROBI) recommended by the Cochrane Handbook for Systematic Reviews of Interventions were chosen.
Data extraction
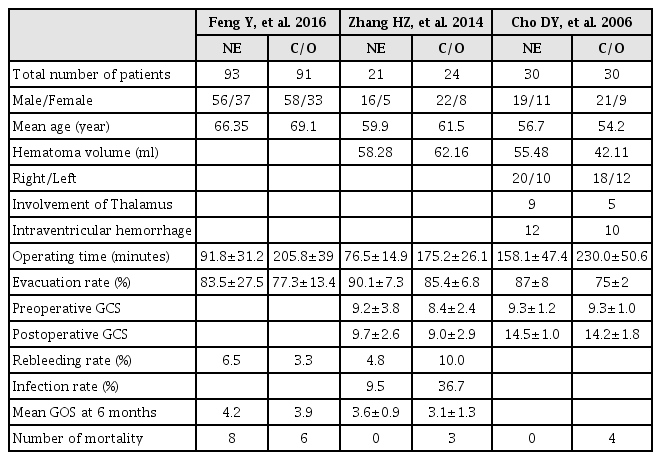
Data were extracted by two authors independently. Disagreement was revolved by consensus. From each eligible study, data of the following were extracted: first author family name, publication year, study total number, mean age, hematoma volume (mL), and death number.
Ethical approval was waived because this study did not involve human participants or animals. Informed consent was not required because no human participant was involved in this study.
Statistical analysis
Meta-analysis was performed by pooling results of reported incidence of death and morbidity of the individual therapeutic modality. Results are expressed as appropriate ratio/difference for dichotomous outcomes based on available data. The I2 statistic test was performed to further examine statistical heterogeneity between trials8). Studies with an I2 ≥ 50% were considered to have moderate and high heterogeneity while those with I2 < 50% were considered to have low heterogeneity9). Random-effect model was adopted if I2 >50%. Otherwise, fixed-effect model was chosen.
Statistical significance was considered when P value was less than 0.05. All meta-analyses were performed using Review Manager Version 5.3 software (Revman; The Cochrane collaboration Oxford, United Kingdom). Findings of our meta-analysis are shown in forest plots. The risk of bias was evaluated using Begg's test and Egger's test.
RESULTS
Overview of literature search and study characteristics
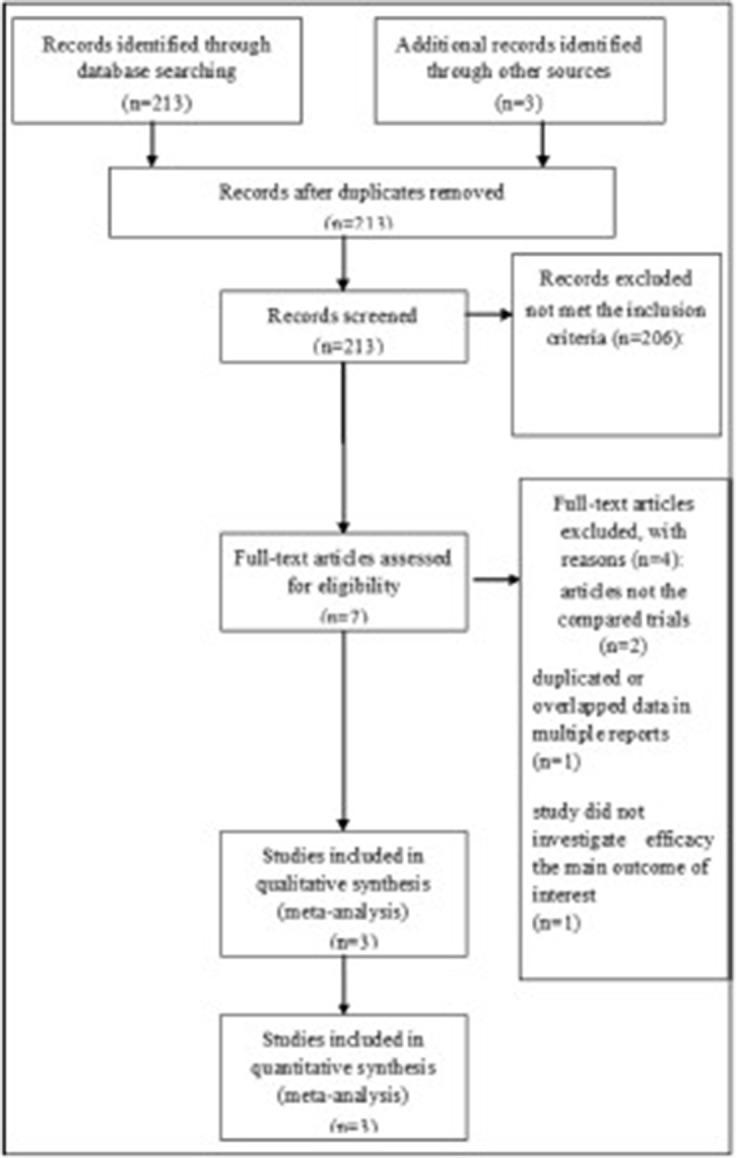
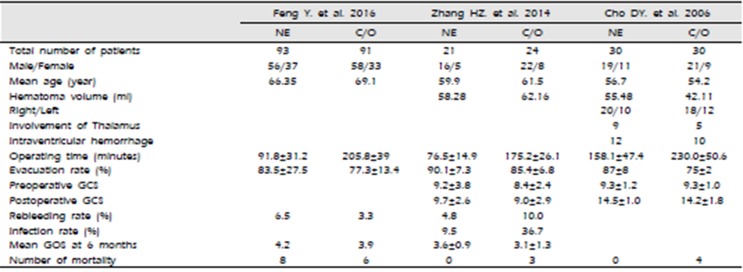
A total of 213 studies were initially found after primary selection. Based on inclusion criteria, 206 irrelevant citations were excluded after reviewing titles and abstracts. Finally, a total of three RCTs5)6)27) were included in this meta-analysis (Fig. 1). These eligible studies included 289 subjects who underwent neuroendoscopic evacuation of supratentorial HICH and craniotomy with removal of HICH (Table 1). Among them, 144 subjects were treated with neuroendoscopic surgery and 145 subjects were treated with craniotomy and removal of HICH. The mean age was 64.1 years old; 63.4 years old in patients who underwent neuroendoscopic surgery and 64.8 years old in patients who underwent craniotomy (P=0.758). The mean volume of HICH was 53.7 mL; 56.6 mL in patients who underwent neuroendoscopic surgery and 51.0 mL years old in patients who underwent craniotomy (P=0.264). The operating time was 103.4 minutes in patients who underwent neuroendoscopic surgery and 205.7 minutes years old in patients who underwent craniotomy (P=0.019). The mean rate of hematoma evacuation was 81.7%; 85.2% in patients who underwent neuroendoscopic surgery and 78.2% in patients who underwent craniotomy (P=0.184). Preoperative Glasgow Coma Scale (GCS) was 9.17; 9.26 in patients who underwent neuroendoscopic surgery and 8.90 patients who underwent craniotomy (P=0.615). Postoperative GCS was 12.23; 12.52 in patients who underwent neuroendoscopic surgery and 11.89 in patients who underwent craniotomy (P=0.759).
Clinical and methodological heterogeneity
1. Pooled analysis of death after neuroendoscopic surgery compare with craniotomy
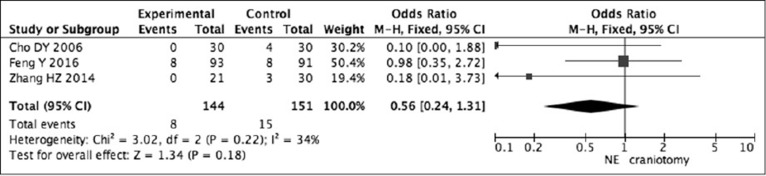
The Odds Ratio (OD) was 0.10 (95% confidence interval [CI]: 0.00–1.88) in Cho's study, 0.98 (95% CI: 0.35–2.72) in Feng's study, and 0.18 (95% CI: 0.01-3.73) in Zhang's study. Pooling analysis5)6)27) revealed that there was no statistically significant difference in death between neuroendoscopic surgery and craniotomy (OR = 0.56, 95% CI: 0.24–1.31; P = 0.18) (Fig. 2).
2. Pooled analysis of complication after neuroendoscopic surgery compare with craniotomy
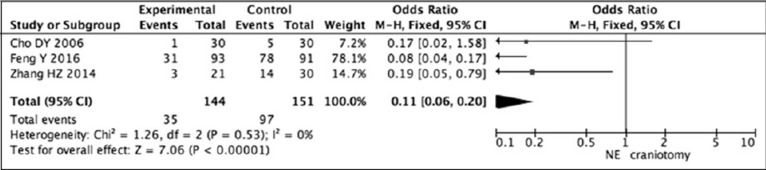
The Odds Ratio (OD) was 0.17 (95% CI: 0.02–1.18) in Cho's study, 0.08 (95% CI: 0.04–0.17) in Feng's study, and 0.19 (95% CI: 0.01–0.79) in Zhang's study. Random-effects model was used to pool complication data5)6)27). Pooled data showed that neuroendoscopic surgery had lower risk of complications than craniotomy (OR = 0.11, 95% CI: 0.06–0.20; P < 0.00001) (Fig. 3).
DISCUSSION
Hypertensive intracerebral hemorrhage is a major health burden with the highest morbidity and mortality. In case of a massive hematoma, neurosurgical drainage is a crucial therapeutic option. Endoscopic evacuation in comparison with traditional craniotomy has been investigated5). However, many questions regarding minimally invasive surgery remain unanswered. Historically, craniotomy has been used as an appropriate therapy to treat HICH4). A recent RCT has shown that early craniotomy surgery might reduce mortality of ICH patients17). Craniotomy also has some advantages such as good view and immediate removal of hematoma with improvement in local blood circulation19) that could improve outcomes. However, there is no more studies showing good results neither any improvement of the outcome in HICH patients who underwent craniotomy and removal of ICH. Because of the lack of large multi-centric RCTs7), many surgical trials have shown that craniotomy is associated with substantial toxicity10)12)20). The shortcoming of craniotomy is that it increases operation time and the risk of infection. Therefore, it is necessary that a minimally invasive surgery causing minimal trauma to normal brain region during the process of removal of hematoma should replace craniotomy to treat HICH13).
During neuroendoscopic surgery as a minimally invasive procedure, a small burr hole is created and a 5 to 8-mm-diameter endoscope is inserted into the brain tissue25). This minimal invasive surgery is performed using an operation endoscope, which has benefit of keeping normal hemostasis of brain and little damage for the skull due to making small bony window compared to craniotomy. Neuroendoscopic surgery has been successfully applied for hematoma evacuation with many advantages28). Some scholars have emphasized that hemorrhage position, hemorrhage volume, and patient condition should be considered in the selection of surgical method11)14). Appropriate operative route is the key to achieve successful treatment of HICH. As neuroendoscopic removal of HICH is preceded within the cavity, it is possible to perform minimally invasive interventions for brain tissues regardless whether these tissues are normal or surrounded by damaged region27).
Regarding the incidence of complications, we found that patients who had undergone neuroendoscopic surgery had fewer complications than those who had undergone craniotomy. The patients who underwent neuroendoscopic evacuation of HICH had lower rate of infectious complications. The reason for this benefit includes the following: (a) neuroendoscopy provides multi-angle observation and “observe around the corner” capability to manage intraoperative bleeding which make up for the insufficiency of direct vision6); and (b) in some studies, to avoid brain tissue damage, some authors have selected short and precise routes to remove hematomas under direct vision and deep lesions without manipulating or exposing unaffected areas27).
Although this meta-analysis showed that the neuroendoscopic surgery should have a benefit compared with the craniotomy for HICH, there is several limitations for application in the practice. Mainly, only three RCTs were eligible for presenting analysis. The parameters were not completely homogeneous. Even several parameters were obtained only one RCT such as anatomical involvement of hemorrhage into the thalamus and ventricle which has meaningful parameter for evaluation of postoperative neurological outcome. And the enrolled patient's age was also different; the Feng's analysis was subjected to the elderly patients with age of 65 to 79 years. The initial neurological status was somewhat different from individual study; the Cho's study was performed in the noncomatose patients. Even the design of Cho's study included patients who underwent stereotactic aspiration. In fact, the parameters for patient's clinical characteristics which were extracted homogenously were only gender, and age of patient. And for the analysis of clinical outcome, the complication and the mortality were parameters which were completely extracted from three RCTs. Therefore, the presenting meta-analysis can show the comparative results in these limited parameters such as postoperative mortality and complications. To overcome above limitation, it is mandatory that further randomized clinical trials should verify the efficacy of neuroendoscopic approach for HICH in the future.
CONCLUSION
Our results suggest that neuroendoscopic surgery can significantly reduce the rate of complications in patients with HICH compared to craniotomy, although it does not improve death outcome. When all data were analyzed, it became obvious that these two methods had their own advantages and shortcomings. Each had its own indications. It is difficult to decide which one is better for HICH patients. Thus, it is essential to select individualized treatment for each patient. Eligible randomized clinical trials are needed to verify the efficacy of neuroendoscopic approach for HICH in the future.
Notes
Funding: No funding was received for this research.
Conflict of Interest: All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.