Iatrogenic Carotid-Cavernous Fistula after Stent Assisted Coil Embolization of Posterior Communicating Artery Aneurysm
Article information
Abstract
Stent assisted coiling (SAC) is a useful technique for the treatment of wide necked complex aneurysm. As the frequency of SAC increases, stent-related complications such as thromboembolism, aneurysm rupture, and vessel rupture have been reported. However, to the best of our knowledge, carotid-cavernous fistula (CCF) after SAC has never been reported. The authors experienced a case of direct CCF after a SAC procedure for treatment of a complex posterior communicating artery (PCoA) aneurysm regrowth, which was treated by clip ligation 12 years before. The patient was managed conservatively and angiograms performed three months after the procedure showed the complete obliteration of the left PcoA aneurysm and the spontaneous disappearance of CCF. Navigation of Solitaire stent lumen with microcatheter can cause unexpected arterial injury, especially when the proximal tip is placed in the curved portion. It seems to be desirable to place the proximal tip of Solitaire stent in the straight portion whenever possible to reduce the risk of inadvertent arterial injury which might be caused by future navigation of stent lumen.
INTRODUCTION
Stent assisted coiling (SAC) has been increasingly performed in patients with wide necked complex aneurysm.1)15)18)27)28) However, the risk of complications increases with the use of stent assistance.14)22)25)30) These complications include thromboembolism, such as parent artery occlusion, distal embolic infarction, aneurysm rupture, and, less frequently, vessel rupture. However, carotid-cavernous fistula (CCF) after SAC has never been reported. In this report, the authors describe a case of direct CCF after a SAC procedure for treatment of a complex posterior communicating artery (PCoA) aneurysm regrowth, which was treated by clip ligation 12 years before.
CASE REPORT
The patient, a 67-year-old woman with a history of spontaneous subarachnoid hemorrhage (SAH) due to a left PCoA aneurysm rupture treated by clip ligation 12 years ago, presented at the emergency department with a sudden severe headache. A computed tomography (CT) scan of the brain showed evidence of a Fisher grade 3 SAH within the basal cisterns and both Sylvian fissures. A cerebral angiogram revealed a posteromedially regrowing, irregularly shaped PCoA aneurysm, with a neck size of 7.22 mm and a dome size of 7.3 × 5.2 mm (Fig. 1). The origin of a small PCoA was incorporated in the aneurysmal neck. Allcock test revealed that the flow to the left posterior cerebral artery was sufficiently maintained from the vertebrobasilar system. We planned a stent-assisted coil embolization to prevent coil loop herniation into the internal carotid artery (ICA) lumen.
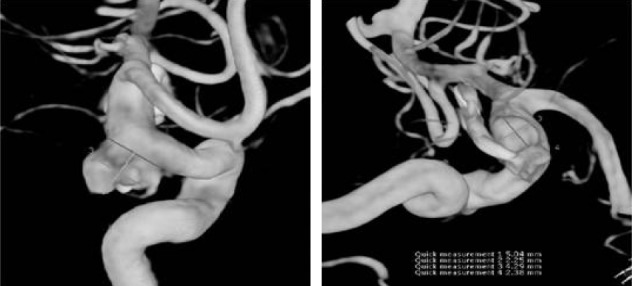
A cerebral angiogram revealed a posteromedially regrowing, irregularly shaped posterior communicating artery (PCoA) aneurysm, with a neck size of 7.22 mm and a dome size of 7.3 × 5.2 mm. The origin of a small PCoA was incorporated in the aneurysmal neck.
Under general endotracheal anesthesia, a 6 French short arterial sheath was placed in the right femoral artery and a 6 French Envoy guiding catheter (Cordis Endovascular, Miami Lakes, FL, USA) was placed at the level of the distal cervical ICA. A Prowler Select Plus microcatheter (Cordis Endovascular, Miami Lakes, FL, USA) and Agility 14 microwire were navigated into the left middle cerebral artery (MCA). Then, we selected the aneurysm sac using a Headway-17 90 degree microcatheter (Microvention Inc., Tustin, CA, USA). Aneurysm coiling was performed using the stent jailing technique. We deployed the Enterprise stent 4.5 mm × 28 mm (Cordis, Miami Lakes, FL, USA) through the Prowler Select Plus microcatheter and did coil packing through a Headway-17 microcatheter. After packing eight coils, it became difficult to verify whether the ICA lumen was compromised by coils (Fig. 2A). Therefore, bailout stenting was performed using a Solitaire stent of 5 mm × 30 mm (ev3 Inc., Irvine, CA, USA). After stenting, the A1 flow was not visualized during another coil packing. We suspected an anterior cerebral artery (ACA) occlusion due to thrombus. Therefore, we decided to perform thrombolysis. In order to advance the microcatheter to A1 through the stent lumen, we had to push and pull the microcatheter multiple times. CCF developed after this action. However, using a contralateral ICA angiogram through another femoral puncture, we determined that it was not a thrombotic occlusion, but a simple flow direction change due to the deployed double overlapping stent across the ACA orifice.
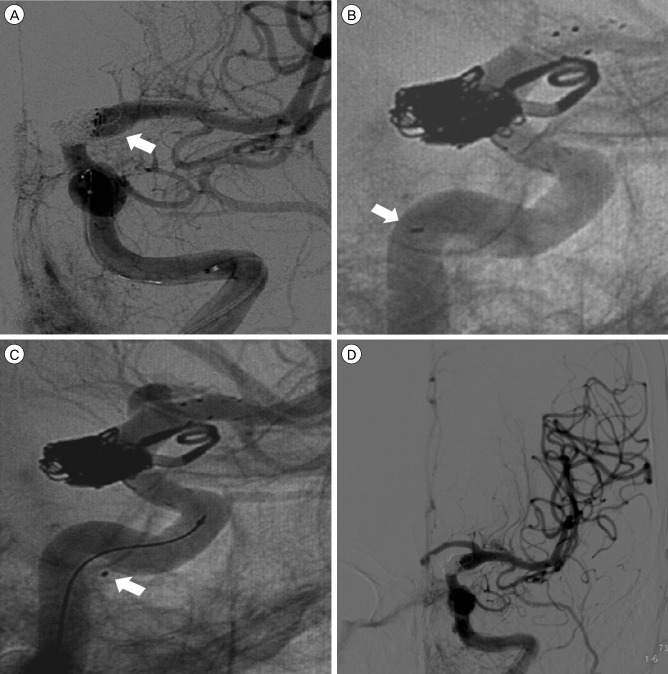
After packing eight coils, it became difficult to verify whether the internal carotid artery (ICA) lumen was compromised by coils (A). Before carotid-cavernous fistula (CCF) occurrence, the proximal tip of the Solitaire stent was located within the ICA lumen (B). Shortly after CCF occurrence, the authors identified the proximal tip of the Solitaire stent beyond the confines of the ICA lumen (C). The procedure was stopped after complete obliteration of the aneurysm without compromising ICA and middle cerebral artery flow, while the CCF remained (D).
The control angiogram showed a small amount of contrast leaking into the cavernous sinus and right inferior petrosal sinus from the cavernous ICA. The proximal tip of the Solitaire stent was visible beyond the confines of the ICA lumen (Fig 2B, C). After packing four more coils, complete obliteration of the aneurysm was achieved without compromising ICA and MCA flow. The procedure was stopped while the CCF remained (Fig. 2D). A postoperative CT scan confirmed no hemorrhagic complication.
After the procedure, the patient was alert and her hospital course was uneventful. A follow-up angiogram seven days after the procedure showed the complete obliteration of the aneurysmal sac and no evidence of vasospasm. However, a high-flow CCF was still visible without the cortical venous reflux, probably due to the perforation of cavernous ICA by the Solitaire stent proximal marker during the procedures (Fig. 3A). The fistula was drained in an antegrade fashion to the inferior petrosal sinus, and the amount of fistulous flow was minimal and localized in the extracranial ICA portion. Furthermore, she had no clinical symptoms of CCF, thus we decided to manage the patient conservatively. Angiograms performed three months after the procedure showed the complete obliteration of the left PcoA aneurysm and the spontaneous disappearance of CCF (Fig. 3B).
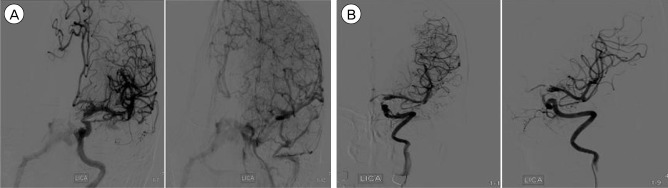
A follow-up angiogram seven days after the procedure. A high-flow carotid-cavernous fistula (CCF) was still visible without the cortical venous reflux (A). After conservative management, angiograms from three months after the procedure showed the complete obliteration of the left posterior communicating artery aneurysm and the spontaneous disappearance of CCF (B).
DISCUSSION
Treatment of aneurysm regrowth after surgical clipping or endovascular coiling is challenging, especially if the shape of the aneurysm is complex or irregular, and the neck is wide. Surgical clipping of previously clipped regrowing aneurysms is challenging because the microsurgical trajectory adheres to the adjacent neurovascular structure, and final clip placement is hindered by the previous clip.2)8)22) Thus, endovascular coiling is a preferable alternative treatment rather than a repeated microsurgical clipping.7)19) In our case, endovascular coiling was selected as an ideal treatment because of the aneurysm's directionality and its irregular shape. The aneurysm projected to the posteromedial direction of the ICA, not to the usual posterolateral direction of PCoA aneurysms. If surgical clipping had been selected for this patient, clip placement over the previous clip might be very difficult and identification of PCoA and its perforators could have been impossible. During the SAC of the PCoA aneurysm, sudden non-visualization of ipsilateral ACA occurred. At that time, we suspected ACA occlusion due to thrombus. However, a contralateral ICA angiogram through another femoral puncture confirmed that it was simply a change in flow direction caused by the stent overlapping the ACA orifice. The cavernous ICA injury developed from the proximal tip of a Solitaire stent while we attempted to advance the microcatheter through the stent lumen. Fortunately, the injured portion of the ICA was the cavernous portion instead of a supraclinoid portion, which could provoke a massive SAH or even intracerebral hemorrhage (ICH).
Direct CCF is usually managed by occlusion of the fistulous portion via a transvenous or transarterial approach.4)5)9) However, in our case, iatrogenic CCF was not treated because the fistula was only drained in an antegrade fashion to the inferior petrosal sinus, and the amount of fistulous flow was estimated to be minimal. Even though it was not a low-flow fistula, we thought there was a high chance of spontaneous occlusion. Furthermore, we expected that the consequences of persistent CCF would be benign because of the absence of intracranial or orbital reflux, even if spontaneous occlusion did not occur within a short term period.6)9)
The Solitaire stent is a new, self-expandable, laser-cut, and fully retrievable split-designed nitinol device which is useful in the treatment of aneurysms with wide necks or unfavorable geometric features.12) Many recent studies have reported on the feasibility and efficacy of the Solitaire stent in treatment of aneurysms at parent vessel bifurcations.10)16)20)23)29) The Solitaire stent system consists of an attached firm push wire that enables the deployment and retrieval of the device (Fig. 4). Although the Solitaire stent is known to be highly flexible, the proximal tip component cannot be flexible due to its structural nature. The proximal tip consists of a fusion of three wires. If this point sticks against the vessel's wall, or if it is pushed against the wall with other devices, the vessel can be injured due to the inflexibility of the proximal tip. In our case, the perforated portion was cavernous ICA, resulting in CCF.
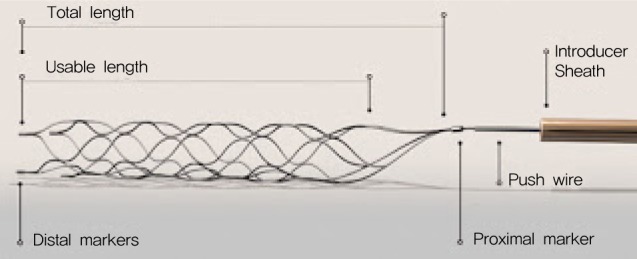
The structure of the Solitaire stent. The stent system consists of an attached firm push wire that enables the deployment and retrieval of the device. Although the Solitaire stent is known to be highly flexible, the proximal tip component cannot be flexible due to its structural nature.
The most common cause of direct CCF is trauma such as blunt head trauma, optic nerve injury and iatrogenic injury during transsphenoidal surgery.3)11) This type of CCF disrupts all layers of the vessel wall exteriorly and extensively, so that is less likely to resolve spontaneously and will require rapid treatment if symptoms occur.24) Vessel wall perforation by a microcatheter or microwire is a rare complication during endovascular treatment. Kwon and Jin reported two cases of direct CCF during microcatheter/microwire navigation that healed spontaneously.13) To the best of our knowledge, this is the first case of CCF provoked by an intraluminal stent. This type of injury disrupts the vascular wall interiorly from the intima to the adventitia. Although the mechanism of spontaneous regression has not yet been elucidated, there is a hypothesis that the iodinated contrast media aggregation promotes clot formation in the focal injured vessel walls, causing spontaneous regression.17)21)26) We believe that iatrogenic CCF can be managed conservatively, especially if the fistulous flow does not involve intracranial and orbital circulation in retrograde fashion.
CONCLUSION
Navigation of Solitaire stent lumen with microcatheter can cause unexpected arterial injury, especially when the proximal tip is placed in the curved portion. It seems to be desirable to place the proximal tip of Solitaire stent in the straight portion whenever possible to reduce the risk of inadvertent arterial injury which might be caused by future navigation of stent lumen.
Notes
Disclosure: The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
