 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 22(2); 2020 > Article |
|
Abstract
Objective
The aim of this study was to assess the feasibility, safety and efficiency of minipterional craniotomy (MPT) for surgical clipping of anterior circulation aneurysms.
Methods
A retrospective study was conducted to compare the MPT from Jan 2015 to Dec 2018 and conventional pterional craniotomy (CPT) from Jan 2012 to Dec 2013 in unruptured intracranial aneurysms (UIA) and ruptured intracranial aneurysms (RIA). The feasibility and safety of MPT and CPT were assessed by analyzing medical records, radiologic imaging, and clinical outcomes. The efficiency of MPT and CPT were based on a survey research of temporomandibular dysfunction, facial nerve paralysis, and facial asymmetry.
Results
Total 628 patients who underwent 458 MPT (UIA:313, RIA:145) and 170 CPT (UIA: 106, RIA: 64) with anterior circulation aneurysms were included in this study. The baseline characteristics between MPT and CPT had no difference (p>0.05). There was no difference in the incidence of postoperative hemorrhage or ischemic lesions between MPT and CPT (p>0.05). The incidence of surgical wound infection was lower in MPT (0.4%) than CPT (3.5%) (p=0.002). More than 90% of postoperative pain disappeared faster in MPT (14.25┬▒4.83 days) than CPT (27.59┬▒10.35 days), and the feeling of facial asymmetry in surgical side was also lower for MPT (1.7%) than CPT (7.6%) (p<0.001). In the MPT, no patients presented with progress to chronic pain, masticatory disability, discomfort of maximal mouth opening or permanent facial palsy.
Conclusions
We suggest that MPT and CPT had similar clinical outcomes, and MPT showed better functional and cosmetic outcomes than CPT in terms of temporomandibular dysfunction, facial nerve paralysis, and facial asymmetry. Therefore, MPT for surgical clipping of anterior circulation aneurysms can be a compatible technique that satisfies the feasibility, safety and efficiency.
Although pterional craniotomy developed by Yasargil in 1975 is the most favored technique to easily approach anterior circulation aneurysms through transsylvian and subfrontal corridors until the present time,29) temporomandibular joint dysfunction and facial asymmetry due to the iatrogenic trauma of temporal muscle are major drawbacks that cannot be denied.1)5)14)19)23)26) Neurosurgeons have taken an interest in these problems, which was included in conventional pterional craniotomy (CPT), and have begun to explore methods that reduce the surgical invasiveness and enhance the patientŌĆÖs quality of life.7)12)21)28)
Various approaches to improve these functional and aesthetic defects have been constantly studied by reducing the size of craniotomy or moving the location of craniotomy to the frontal portion.10)13)18)20)24)25) Among them, CPT-modified minipterional craniotomy (MPT) has been introduced since the early 2010s as an alternative less invasive technique that decreased the length of skin incision, damage of temporal muscle, and craniotomy size.2)6)8)9)11)15-17)22)27) However, some studies did not mention whether the clinical results of CPT were maintained in MPT,2)8)25) and did not sufficiently verify the evaluation of functional efficiency by emphasizing only the lesser invasiveness.6)11)16) Therefore, we performed a study to assess the feasibility, safety and efficiency of MPT for surgical clipping of unruptured intracranial aneurysms (UIA) and ruptured intracranial aneurysms (RIA), with a special focus on the specific indication in the individual type of anterior circulation aneurysms.
After the approval of institutional review board (IRB), the authors conducted a retrospective analysis of all patients with anterior circulation aneurysms (n=1,068) at our hospital between January 2015 and December 2018. All aneurysms were first treated with endovascular coiling if possible (n=543). Thus, the remaining aneurysms not suitable for coiling were subjected to surgical clipping (n=525). During the study period, surgical clipping of most anterior circulation aneurysms was performed with MPT. Although MPT was launched in January 2014, the cases of first year were not included in the population considering the skill and familiarity of operator. Patients who underwent wide craniotomy or craniectomy due to RIA with large volume ICH (n=23), performed parasagittal craniotomy for pericallosal aneurysm (n=18), had undergone a previous craniotomy (n=11), had a history of facial palsy (n=0), or failed to get a follow-up radiological examination and appointment at outpatient department (OPD) for over 6 months (n=15) were excluded from the study population. Therefore, 458 patients with 562 aneurysms were included in the MPT group.
We also conducted a retrospective review of patients between January 2012 and December 2013 to compare the outcomes of MPT and CPT (n=274). Population criteria for CPT were the same as those for MPT. Thus, 170 patients with 198 aneurysms were included in the CPT group. All surgical procedures in the two groups were performed by one senior neurosurgeon (C.Y. Lee). Both UIA (MPT: 313, CPT: 106) and RIA (MPT: 145, CPT: 64) were included in our study. For the UIAs, transfemoral cerebral angiography (TFCA) was performed to evaluate the number, location, size and shape of aneurysms. Scheduled surgery was performed based on the patientŌĆÖs age and medical history after a preoperative risk assessment. For the RIAs, emergency surgery was performed within 1 day of the hospital visit in all cases after taking a computed tomographic angiography (CTA).
A curvilinear scalp incision was made from the height of lateral epicanthus to the ipsilateral midpupillary line. The skin incision was made just posterior to hairline, and it was located anterior to incision in CPT. the incision line was made such that the scar would be completely covered by patientŌĆÖs hair after recovery. Thus, the length of the MPT incision was shorter than that of the CPT incision, especially in patients with a smaller head and narrower forehead. The patientŌĆÖs head was only shaved up to 2-3 cm posterior to the incision line, in order to reduce the patientŌĆÖs discomfort (Fig. 1).
Infero-lateral to superior temporal line (STL), the scalp flap was reflected through performing subfascial dissection between the deep temporal fascia and temporalis muscle, in order to minimize exposure of the superficial temporal fat pad, which contains the frontotemporal branch of the facial nerve. Supero-medial to STL, the galea aponeurotica, loose areolar tissue, and pericranium were left to the skull so that re-approximation could be performed by suturing with the temporalis fascia at the end of the surgery. An incision was made in the temporalis fascia along the STL, and a subperiosteal dissection of the temporal muscle was performed to expose the pterion using a dura elevator, rather than monopolar cautery.
The upper border of the craniotomy was within the inferior range of the STL. Craniotomy was performed in a downward curve towards the squamous temporal bone using the pterion as a distal landmark in MPT, unlike CPT, where the pterion is a central landmark. Therefore, MPT included the anterior part of the frontal bone inferior to the STL, the lateral part of the sphenoid bone, and only a minimal portion of the squamous temporal bone. The size of MPT was much reduced to 2.5├Ś2 cm or 3├Ś2 cm. Burr-hole was performed only once on the superoposterior side of the bone flap to minimize the temporal hollowing. The outer table of skull was preserved to the maximal extent during the sphenoid ridge drilling. These processes of craniotomy were performed only beneath the temporal muscle (Fig. 2).
When a semilunar dural incision was made on the base of the flattened sphenoid ridge, part of the inferior frontal gyrus, the anterior ascendant ramus of the Sylvian fissure, and part of the superior temporal gyrus were exposed (Fig. 3). Sufficient microsurgical field could be obtained by cerebrospinal fluid (CSF) drainage using an arachnoid dissection and frontal or temporal retraction using a Greenberg retractor (Fig. 4). In the RIA, an intraoperative ventriculostomy was performed at PaineŌĆÖs point (the point of an isosceles triangle with the Sylvian fissure as the base and a side-length of 2.5 cm) to alleviate brain swelling (Fig. 5).
After aneurysmal clipping has been completed, the bone flap was fixed in anatomical alignment using cranial plate hardware consisting of a burr-hole cover and two straight bars, autologous bone chips and hydroxyapatite cements to fill the gap (Fig. 6). The incised temporalis fascia and the galea aponeurotica and pericranium, which had been left on the superomedial side of the STL, were reapproximated as tightly as possible using Vicryl absorbable suture. And then the craniotomy scar was completely concealed by the temporal muscle (Fig. 7).
To assess the feasibility of MPT in anterior circulation aneurysms, the sex, age, presence of hemorrhage, and operation time were compared with CPT. The size, location and direction of aneurysm, and the relationship between skull and aneurysm were examined by analyzing preoperative CTA and TFCA. Additionally, the applicability of MPT in the case of subarachnoid hemorrhage (SAH) due to RIA was investigated by examining Hunt and Hess (H-H) grade and FisherŌĆÖs scale.
To examine the microsurgical safety of MPT and CPT, postoperative radiologic data and medical records were analyzed. We confirmed the presence of postoperative ICH or ischemic lesions, and epidural (EDH) or subdural hematoma (SDH) on postoperative immediate and 8th day computed tomography (CT). In the RIA, the presence of vasospasm or delayed hydrocephalus was verified, and the presence of aneurysmal remnants was confirmed through TFCA. we checked the presence of surgical wound infection, cerebrospinal fluid (CSF) leakage, meningitis and brain abscess. Permanent neurological deficits were also evaluated with the modified Rankin scale (mRS) score.
To assess the functional and cosmetic efficiency of MPT and CPT, we analyzed temporomandibular dysfunction, facial nerve paralysis, facial asymmetry, and surgical aesthetic results. A survey research was carried out in all patients at about 6 months after surgery, and the questionnaire included the following 11 items. In order to verify postoperative temporomandibular joint dysfunction, the survey evaluated time durations for postoperative pain to disappear, progression to chronic pain, masticatory disability, and discomfort of maximal mouth opening. To identify the occurrence of facial nerve paralysis, we investigated drooping of the upper eyelid, duration of ptosis, progression to permanent ptosis, and unilateral facial weakness. In addition, the change of facial sense due to skin incision, and facial asymmetry due to temporal muscle atrophy were examined. The subjective satisfaction of patients was scored of 0-10 based on a combination of the above questions.
SPSS 20.0 for Windows (SPSS Inc., Chicago, IL, USA) was used for the statistical analyses in this study. The clinical outcomes and complications between MPT and CPT were respectively analyzed by examining several variables such as postoperative hemorrhage or infarction, surgical infection, vasospasm and hydrocephalus. The functional outcomes of MPT and CPT were based on a survey research (including 11 questionnaires) of temporomandibular dysfunction, facial nerve paralysis, facial asymmetry, and patientŌĆÖs satisfaction in all patients. The FisherŌĆÖs exact test and the independent t-test were performed for the statistical significance between two groups. Results with p-values less than 0.05 were considered statistically significant.
The baseline characteristics of patients consisting of 458 cases of MPT (UIA: 313, RIA: 145) and 170 cases of CPT (UIA: 106, RIA: 64) are summarized in Table 1. There were no significant differences between the two groups in sex, age, size and location of aneurysm, presence of hemorrhage, and H-H grade and FisherŌĆÖs scale for RIA (p>0.05). 562 anterior circulation aneurysms were surgically treated with MPT. The mean size of these aneurysms was 4.56┬▒2.03 mm, and 28 aneurysms above 10 mm in size and 87 cases of multiple aneurysms were treated without a problem. The operation time for MPT (154.66┬▒39.31 minutes) was about 54.0 minutes shorter than that for CPT (208.65┬▒35.62 minutes) (p<0.001). 18 patients underwent bilateral MPT under a single round of anesthesia, and these cases were excluded from the comparison of operation time.
We treated 256 middle cerebral artery (MCA) aneurysms with a mean length of 16.52┬▒5.46 mm from the internal cerebral artery (ICA) bifurcation. We were able to obliterate aneurysms from MCA M1 located at medial position of 2.93 mm (n=31) to MCA bifurcation located at the lateral position of 30.56 mm (n=225). There were 154 anterior communicating artery (ACoA) aneurysms. The mean height of the ACoA aneurysmal neck was 6.52┬▒2.67 mm, and aneurysms with a height up to 14.69 mm could be obliterated. The aneurysm height was defined as the vertical distance from the planum sphenoidale to the highest point of the neck as observed on sagittal CT images. Aneurysmal neck with superior or posterior direction occluded by the ipsilateral anterior cerebral artery (ACA) A2 was clipped through the contralateral approach (n=15). We treated 62 posterior communicating artery (PCoA) aneurysms regardless of the direction of lesion. The distance between the origin of ophthalmic artery and the lesion was 13.15┬▒2.49 mm. Extradural anterior clinoidectomy could be performed in 8 cases when temporary clipping was not possible because the length was shorter than 10 mm and anterior clinoid process (ACP) was large. We also clipped 57 anterior choroidal artery (AchA), 18 ACA A1, 4 ACA proximal A2, and 11 ICA bifurcation aneurysms with MPT (Table 2).
Only 4 cases of intraoperative bleeding were observed during aneurysmal clipping with MPT, 2 unruptured MCA bifurcations and 2 unruptured PCoA aneurysms. However, temporary clipping was applied with rapid suction of the squirted blood, and the aneurysm was obliterated safely. Hemorrhage was not found on postoperative CT images, and the patients showed no other symptoms. There was no case in which the MPT had to be converted to CPT because of an insufficient microsurgical field in dealing with aneurysms. Postoperative TFCA was performed for the assessment of aneurysmal remnants in all RIA cases in MPT (n=145). Aneurysmal remnants after surgical clipping were found in a ruptured 3 ACoA and 1 PCoA aneurysms (2.8%). However, all four of cases were followed up without re-surgery as only a slight portion of the aneurysmal neck remained, and no rebleeding occurred before the final OPD follow-up.
There was no significant difference in the incidence of postoperative hemorrhage or ischemic lesions between MPT and CPT (p>0.05). Two cases required surgical evacuation, one for a frontal lobe ICH in MPT, and one for a basal ganglia ICH in CPT. The incidence of surgical wound infection was lower in MPT (0.4%) than in CPT (3.5%) (p=0.002). Incidence of CSF leakage and meningitis or brain abscess was not occurred in MPT. There was no significant difference in the symptomatic vasospasm and delayed hydrocephalus between the two groups (p>0.05) (Table 3).
MPT for surgical clipping of UIA was not associated with postoperative reduction in mRS score, or with permanent neurological deficits. In the RIA, there were 1 deaths of patients within 3 months caused by Acinetobacter baumannii meningitis due to prolonged lumbar CSF drain, and 4 deaths within 6 months due to internal medicine problems. Except for 5 deaths unrelated to surgical technique of MPT, which were caused by infection or other medical problems, successful clipping was performed in 145 patients with an RIA, including 109 cases with a Fisher scale of 3-4.
The time required for over 90% of the postoperative jaw pain to disappear was about 13.3 days fewer for MPT (14.25┬▒4.83 days) than for CPT (27.59┬▒10.35 days) (p<0.001). Progression to chronic pain, masticatory difficulties and discomfort of maximal mouth opening were not seen for MPT. Although postoperative ptosis was less prevalent for MPT (7.0%) than for CPT (14.1%) (p=0.005), the temporary incidence itself appeared somewhat high in both the groups. However, no cases of permanent unilateral facial weakness occurred for MPT, including ptosis. The rate of skin numbness at surgical site was lower for MPT (5.9%) than CPT (13.5%) (p=0.002), and the rate of facial asymmetry between surgical side and non-surgical side was also lower for MPT (1.7%) than for CPT (7.6%) (p<0.001). After combining the above items, the subjective satisfaction of the patient was also found to be higher for MPT (9.03┬▒1.15) than for CPT (7.66┬▒1.43) (p<0.001) (Table 4).
We made effort to improve the MPT for aneurysmal clipping by modifying the surgical techniques such as decreasing the length of skin incision, preserving the temporal muscle, and reducing the craniotomy size. First, the shortened incision line was able to reduce skin numbness at the surgical site by about 7.4%. Of course, the length could be shortened further if a straight skin incision were made at the craniotomy location in the pterional area. Some studies have attempted this,16) but because the tiny scar over this area is on an exposed part of the face, it can be more stressful for the patient. Moreover, there is a risk of injury to the frontal branch of facial nerve, and if a problem arises, it could make conversion to CPT more challenging. Therefore, we decided to make the incision just posterior to the hairline so that the scar would be completely covered once the hair grows.
Second, we performed subfascial dissection between the deep temporal fascia and temporalis muscle, which can secure a broader space and prevent facial palsy. The integrity of the muscle fiber was maintained by making an incision in the temporalis fascia and minimizing the use of monopolar cautery. In addition, adequate tension was applied during closure by tightly reattaching the muscle to the galea aponeurotica and the pericranium, which were left on the superomedial side of the STL. Myocutaneous flap, another method to reflect the scalp flap and expose the skull, can completely avoid the exposure of facial nerve, but this requires a long incision of temporal muscle,19)23)30) Above all, it is unsuitable for ACoA aneurysms located in deep positions, since the bulky flap obscures the microsurgical field in MPT.
Third, the size of bone flap was considerably reduced as far as possible to minimize the exposure of cortical areas, which was irrelevant to the surgery. Since the range of surgical field was smaller and the time duration of surgery was reduced by approximately 55.8 minutes, we consider it can be a cause of lower incidence of surgical wound infection. Owing to the reduced physical burden of the surgeon, 18 patients were able to undergo bilateral MPT under one anesthesia. Above all, although the smaller operative window in MPT may seem restrictive, it was also well preserved like the surgical corridor of CPT. Figueiredo et al. have reported that when the pterion is used as a distal landmark for MPT, dissection is performed only up to the anterior ascendant ramus of the Sylvian fissure.9) Any additional opening provides no advantage for inspecting the basal cistern and the circle of Willis, and extensive dissection using the CPT method can rather promote chronic SDH. In other words, the authors inferred that MPT could be applied broadly to anterior circulation aneurysms.
Owing to limitations in exposure of the posterior Sylvian fissure for MCA aneurysms located in a lateral position, MPT can be considered unsuitable in this location. However, if the site of craniotomy is appropriately adjusted through the analysis of distance from the ICA bifurcation and relationship between skull and aneurysm, MPT can be broadly applied in MCA regardless of the direction of aneurysm. In case of long M1 (Ōēź20 mm) (n=71), MCA bifurcation aneurysms could be handled without the need for the conversion to CPT, making only a slight modification to the craniotomy range by shifting it further posteriorly along the axis of the Sylvian fissure. We assume that the lesion at a lateral position of more than 30.56 mm beyond the range of our population can be applied safely.
Although there are studies that exclude ACoA aneurysms located in deep positions because they believe MPT to be unsuitable for these cases,2)6) we considered that MPT would need to be applicable even to ACoA aneurysms, which are known to be highly prevalent and to rupture even at small sizes. Therefore, we were able to successfully obliterate 154 cases with various directions, sizes, and locations. In highly located ACoA aneurysm from the skull base and ACA proximal A2 aneurysms, sufficient surgical field was secured through the proper dissection of interhemispheric fissure. Aneurysm with superior or posterior direction is often occluded by ipsilateral ACA A2, unlike aneurysm with inferior or anterior direction. In this case of requiring more frontal retraction, the contralateral approach was used because MPT could not perform the same retraction as CPT (n=15).
There was concern that anterior clinoidectomy for temporary clipping would be limited by reduced operative window of MPT if the length between the PCoA aneurysm and the origin of ophthalmic artery was short. However, MPT did not differ from CPT in drilling ACP, and the temporary clip mounted on ICA did not interfere with the view of lesion (n=8). In addition, the fact that 130 cases of supraclinoid ICA aneurysms involving the PCoA, AchA, and ICA bifurcation were ligated without a problem, proved that MPT has a broad range of feasibility.
We applied MPT not only to UIA but also RIA. Although most studies have not attempt in RIA with severe cerebral edema,16)22) it was performed as an emergency surgery within 24 hours of the hospital visit in all cases, after only CTA imaging. This was because clipping in a timely manner is the most significant principle in RIA to prevent rebleeding, for induced hypertension to prevent vasospasm, and for earlier initiation of rehabilitation. Therefore, early surgery was performed like in CPT in order not to harm clinical outcomes. Brain swelling could be alleviated sufficiently by CSF drainage from the frontal horn of the lateral ventricle via PaineŌĆÖs point. Ventriculostomy could be enough performed in all cases in which a 3├Ś2 cm craniotomy was performed just to expose this point. Since the increased intracranial pressure (IICP) control is possible in the same way as the CPT, MPT can be applied to RIA even in fisher scale 3-4 with cerebral edema (n=109).
Several reports with MPT for anterior circulation aneurysms included relatively small volume less than 50 cases,8)22)25)27) and we think that the evaluation of clinical and functional results maybe not fully investigated.2)6)11)16) By the way, Rychen et al. reported a systemic review of the literatures on minimally invasive alternative approaches to CPT,21) and they collectively reviewed approach-related complications in a total of 22 MPT articles. Postoperative hematoma and infarction occurred in 2.2% and 3.1% of all patients, and wound infection and CSF leak was reported in 1.2% and 0.3%. Painful mastication occurred in 7.5% of all patients, and permanent facial palsy and temporal hollowing was reported in 0.4% and 0.9%, respectively. The present study showed similar or slightly better results than this systemic review.
However, the incidence of facial palsy-induced ptosis, a longstanding problem of CPT, was somewhat high (7.0%) compared to other functional problems in our study. It has been reported that its incidence could be up to 30% in CPT.3) Considering that the ptosis was temporary and disappeared without any permanent remnants, it was estimated that excessive tension during temporalis muscle retraction was the cause of ptosis. As 1.6% of patients felt facial asymmetry even in MPT, this method could not essentially avoid the bony degeneration and muscle atrophy, so it might be difficult to completely prevent the temporal depression. Therefore, a small amount of hydroxyapatite cement was particularly used for cranial reconstruction, only in cases of women in whom the temporalis muscle and pterional portion of skull were thin. It is possible that excessive frontal retraction or rectus gyrus excision in the ACoA aneurysm located in the deep position may cause anosmia dysfunction,4) unfortunately, but we did not perform an evaluation of postoperative olfactory function. Our study has also some limitations in that it is a retrospective analysis that MTP and CPT were performed in different period, respectively. In addition, since our population was selected based on patients who had OPD follow-up for more than 6 months, we think it might be somewhat short to assess the facial asymmetry. Maybe, long term and large prospective study could be needed in future.
We minimized the injury of temporal muscle and the size of craniotomy for improving the pitfalls of CPT, and in our knowledge, we collected large population among several studies on MPT for aneurysmal clipping at a single institute. The present study showed that overall functional outcomes of MPT were superior to those of CPT, while the clinical outcomes of MPT including postoperative complications were similar to those of CPT. Since no perfect surgical technique exists that can be applied to all cases, it could be necessary that surgeons would know a variety of approaches. However, this requires a long time and considerable effort. Given that MPT is only a slight modification of CPT, which is the most familiar approach for neurosurgeons, it has the potential to become a safe and efficient alternative for surgical clipping in anterior circulation aneurysms.
We reduced the length of skin incision, temporal muscle injury, and size of craniotomy for overcoming the drawbacks of CPT. Although the feasibility of MPT could not be completely the same as CPT, MPT for surgical clipping could be applied not only to UIA, but also to RIA in most anterior circulation. In addition, MPT and CPT had similar clinical outcomes, and MPT showed better functional outcomes than CPT in terms of temporomandibular dysfunction, facial nerve paralysis, and facial asymmetry. Therefore, MPT for surgical clipping of anterior circulation aneurysms can be a compatible technique that satisfies the feasibility, safety and efficiency.
REFERENCES
1. Agarwal CA, Mendenhall SD 3rd, Foreman KB, Owsley JQ. The course of the frontal branch of the facial nerve in relation to fascial planes: an anatomic study. Plast Reconstr Surg. 2010 2;125(2):532-7;


2. Alkhalili KA, Hannallah JR, Alshyal GH, Nageeb MM, Abdel Aziz KM. The minipterional approach for ruptured and unruptured anterior circulation aneurysms: Our initial experience. Asian J Neurosurg. 2017 Jul-Sep;12(3):466-74;



3. Ammirati M, Spallone A, Ma J, Cheatham M, Becker D. An anatomicosurgical study of the temporal branch of the facial nerve. Neurosurgery. 1993 12;33(6):1038-43; discussion 44.


4. Aydin IH, Kadioglu HH, Tuzun Y, Kayaoglu CR, Takci E, Ozturk M. Postoperative anosmia after anterior communicating artery aneurysms surgery by the pterional approach. Minim Invasive Neurosurg. 1996 9;39(3):71-3;



5. Babakurban ST, Cakmak O, Kendir S, Elhan A, Quatela VC. Temporal branch of the facial nerve and its relationship to fascial layers. Arch Facial Plast Surg. 2010 Jan-Feb;12(1):16-23;


6. Caplan JM, Papadimitriou K, Yang W, Colby GP, Coon AL, Olivi A, et al. The minipterional craniotomy for anterior circulation aneurysms: initial experience with 72 patients. Neurosurgery. 2014 6;10(Suppl 2):200-6; discussion 206-7.



7. Davies JM, Lawton MT. Advances in open microsurgery for cerebral aneurysms. Neurosurgery. 2014 2;74(Suppl 1):S7-16;



8. Dzhindzhikhadze RS, Dreval ON, Lazarev VA, Kambiev RL. Minipterional craniotomy in surgery for anterior circle of Willis aneurysms. Zh Vopr Neirokhir Im N N Burdenko. 2016 80(6):-58; -65.

9. Figueiredo EG, Deshmukh P, Nakaji P, Crusius MU, Crawford N, Spetzler RF, et al. The minipterional craniotomy: technical description and anatomic assessment. Neurosurgery. 2007 11;61(5 Suppl 2):256-64; discussion 64-5.


10. Figueiredo EG, Deshmukh V, Nakaji P, Deshmukh P, Crusius MU, Crawford N, et al. An anatomical evaluation of the mini-supraorbital approach and comparison with standard craniotomies. Neurosurgery. 2006 10;59(4 Suppl 2):ONS212-20.discussion ONS20.



11. Figueiredo EG, Welling LC, Preul MC, Sakaya GR, Neville I, Spetzler RF, et al. Surgical experience of minipterional craniotomy with 102 ruptured and unruptured anterior circulation aneurysms. J Clin Neurosci. 2016 5;27:34-9;


12. Fischer G, Stadie A, Reisch R, Hopf NJ, Fries G, Bocher-Schwarz H, et al. The keyhole concept in aneurysm surgery: results of the past 20 years. Neurosurgery. 2011 3;68(1 Suppl Operative):45-51; discussion 51.



13. Hernesniemi J, Ishii K, Niemela M, Smrcka M, Kivipelto L, Fujiki M, et al. Lateral supraorbital approach as an alternative to the classical pterional approach. Acta Neurochir Suppl. 2005 94:17-21;


14. Kadri PA, Al-Mefty O. The anatomical basis for surgical preservation of temporal muscle. J Neurosurg. 2004 3;100(3):517-22;


15. Mocco J, Komotar RJ, Raper DM, Kellner CP, Connolly ES, Solomon RA. The modified pterional keyhole craniotomy for open cerebrovascular surgery: a new workhorse? J Neurol Surg A Cent Eur Neurosurg. 2013 11;74(6):400-4;



16. Mori K, Esaki T, Yamamoto T, Nakao Y. Individualized pterional keyhole clipping surgery based on a preoperative three-dimensional virtual osteotomy technique for unruptured middle cerebral artery aneurysm. Minim Invasive Neurosurg. 2011 10;54(5-6):207-13;



17. Mori K, Wada K, Otani N, Tomiyama A, Toyooka T, Takeuchi S, et al. Keyhole strategy aiming at minimizing hospital stay for surgical clipping of unruptured middle cerebral artery aneurysms. J Neurosurg. 2018 4 1 1-8.

18. Park J. Supraorbital Keyhole Approach for Intracranial Aneurysms: Transitioning from Concerns to Confidence. J Korean Neurosurg Soc. 2020 1;63(1):4-13;



19. Poblete T, Jiang X, Komune N, Matsushima K, Rhoton AL Jr. Preservation of the nerves to the frontalis muscle during pterional craniotomy. J Neurosurg. 2015 6;122(6):1274-82;


20. Reisch R, Marcus HJ, Hugelshofer M, Koechlin NO, Stadie A, Kockro RA. PatientsŌĆÖ cosmetic satisfaction, pain, and functional outcomes after supraorbital craniotomy through an eyebrow incision. J Neurosurg. 2014 9;121(3):730-4;


21. Rychen J, Croci D, Roethlisberger M, Nossek E, Potts M, Radovanovic I, et al. Minimally invasive alternative approaches to pterional craniotomy: a systematic review of the literature. World Neurosurg. 2018 5;113:163-79;


22. Sturiale CL, La Rocca G, Puca A, Fernandez E, Visocchi M, Marchese E, et al. Minipterional craniotomy for treatment of unruptured middle cerebral artery aneurysms. A single-center comparative analysis with standard pterional approach as regard to safety and efficacy of aneurysm clipping and the advantages of reconstruction. Acta Neurochir Suppl. 2017 124:93-100;


23. Tayebi Meybodi A, Lawton MT, Yousef S, Sanchez JJG, Benet A. Preserving the facial nerve during orbitozygomatic craniotomy: surgical anatomy assessment and stepwise illustration. World Neurosurg. 2017 9;105:359-68;


24. Toyooka T, Wada K, Otani N, Tomiyama A, Takeuchi S, Tomura S, et al. Potential risks and limited indications of the supraorbital keyhole approach for clipping internal carotid artery Aneurysms. World Neurosurg X. 2019 2;2:100025



25. Tra H, Huynh T, Nguyen B. Minipterional and supraorbital keyhole craniotomies for ruptured anterior circulation aneurysms: experience at single center. World Neurosurg. 2018 1;109:36-9;


26. Vaca EE, Purnell CA, Gosain AK, Alghoul MS. Postoperative temporal hollowing: Is there a surgical approach that prevents this complication? A systematic review and anatomic illustration. J Plast Reconstr Aesthet Surg. 2017 3;70(3):401-15;


27. Welling LC, Figueiredo EG, Wen HT, Gomes MQ, Bor-Seng-Shu E, Casarolli C, et al. Prospective randomized study comparing clinical, functional, and aesthetic results of minipterional and classic pterional craniotomies. J Neurosurg. 2015 5;122(5):1012-9;


28. Wong JH, Tymianski R, Radovanovic I, Tymianski M. Minimally invasive microsurgery for cerebral aneurysms. Stroke. 2015 9;46(9):2699-706;


Fig.┬Ā1
A curvilinear scalp incision was made from the height of the lateral epicanthus to the ipsilateral midpupillary line, such that the incision line was just posterior to hairline. The patientŌĆÖs head was only shaved up to 2-3 cm posterior to the incision line.

Fig.┬Ā2
The scalp flap was reflected using a subfascial dissection, and an incision was made in the temporalis fascia along the superior temporal line (STL). The craniotomy was performed inferior and lateral to the STL, using the pterion as a distal landmark, and burr-hole (red dotted circle) was performed only once on the superoposterior side of the bone flap.
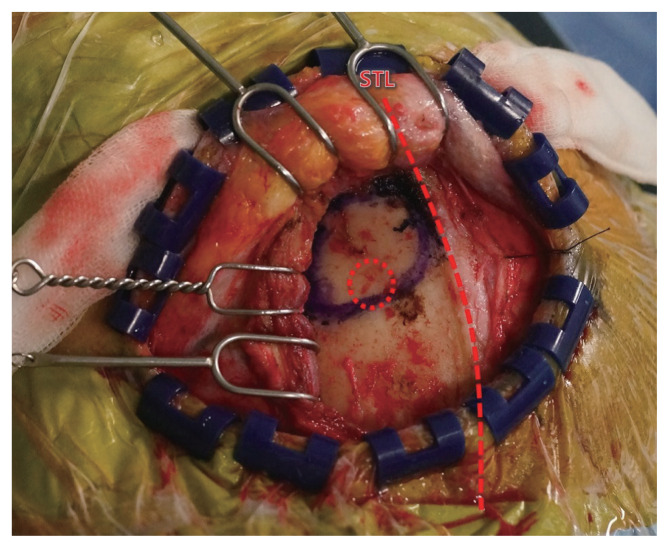
Fig.┬Ā3
After sphenoid ridge drilling and semilunar dural incision, the anterior ascendant ramus of Sylvian fissure, and part of the inferior frontal gyrus and superior temporal gyrus were partially exposed.

Fig.┬Ā4
Unruptured anterior choroidal artery (AchA) aneurysms in a 58-year-old female patient were exposed after securing the microsurgical field through frontal retraction using a Greenberg retractor, and aneurysmal clipping has been completed.

Fig.┬Ā5
Fisher scale 4 hemorrhage was observed in a 72-year-old female patient, due to a ruptured anterior communicating artery (ACoA) aneurysm. The brain swelling was alleviated by cerebrospinal fluid (CSF) drainage through PaineŌĆÖs point, and 3├Ś2 cm craniotomy was performed just to expose this point.
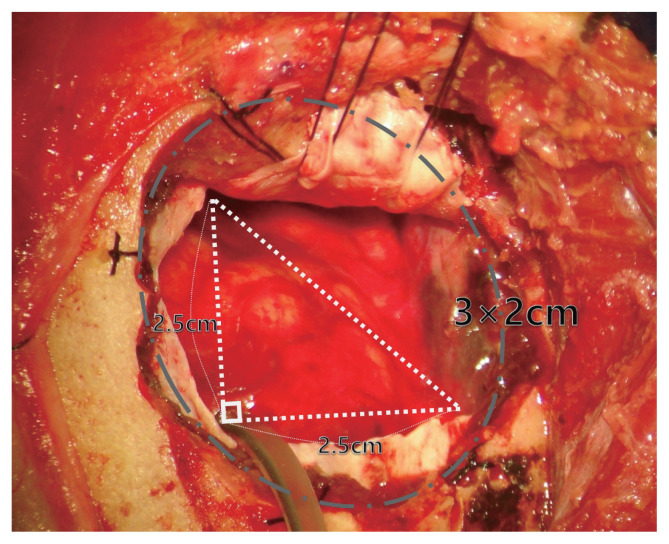
Fig.┬Ā6
The 2.5├Ś2 cm sized bone flap was fixed appropriately using cranial plate hardware consisting of a burr-hole cover and two straight bars.
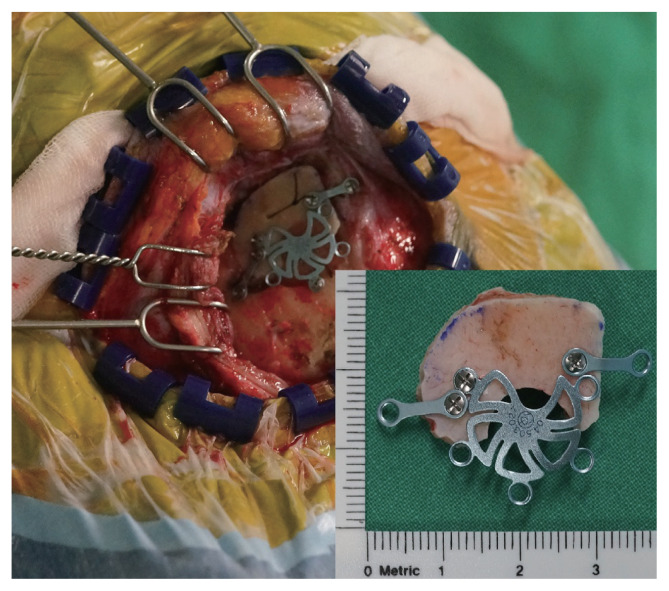
Fig.┬Ā7
The craniotomy scar was completely concealed by reattaching the temporal muscle and the incised temporalis fascia under adequate tension.

Table┬Ā1
Baseline characteristics of 628 patients with 458 MPT and 170 CPT of anterior circulation aneurysms
| Variables | No. of patients (%) | p value | |
|---|---|---|---|
|
|
|||
| MPT (n=458) | CPT (n=170) | ||
| Sex | 0.574 | ||
| ŌĆāMale | 164 (35.8) | 65 (38.2) | |
| ŌĆāFemale | 294 (64.2) | 105 (61.8) | |
| Age (years) | 59.60┬▒9.83 | 58.51┬▒10.66 | 0.235 |
| Size of aneurysm (mm) | |||
| ŌĆā<5 | 359 (63.9) | 117 (59.1) | 0.231 |
| ŌĆā5-10 | 175 (31.1) | 73 (36.9) | 0.139 |
| ŌĆā>10 | 28 (5.0) | 8 (4.0) | 0.592 |
| Location of aneurysm | |||
| ŌĆāMCA | 256 (45.6) | 98 (49.5) | 0.339 |
| ŌĆāACoA | 154 (27.4) | 52 (26.3) | 0.756 |
| ŌĆāPCoA | 62 (11.0) | 22 (11.1) | 0.976 |
| ŌĆāAchA | 57 (10.1) | 19 (9.6) | 0.826 |
| ŌĆāEtc. | 33 (5.9)* | 7 (3.5) | 0.205 |
| Presence of hemorrhage | 0.086 | ||
| ŌĆāUIA | 331 (69.5) | 106 (62.4) | |
| ŌĆāRIA | 145 (30.5) | 64 (37.6) | |
| H-H grade in RIA | 0.454 | ||
| ŌĆā1-3 | 99 (68.3) | 47 (73.4) | |
| ŌĆā4-5 | 46 (31.7) | 17 (26.6) | |
| Fisher scale in RIA | 0.645 | ||
| ŌĆā1-2 | 36 (24.8) | 14 (21.9) | |
| ŌĆā3-4 | 109 (75.2) | 50 (78.1) | |
| Operation time (min) | 154.66┬▒39.31 | 208.65┬▒35.62 | <0.001 |
MPT, minipterional craniotomy; CPT, conventional pterional craniotomy; MCA, middle cerebral artery; ACoA, anterior communicating artery; PCoA, posterior communicating artery; AchA, anterior choroidal artery; UIA, unruptured intracranial aneurysm; RIA, ruptured intracranial aneurysm; H-H, Hunt and Hess; ACA, anterior cerebral artery; ICA, internal carotid artery
Table┬Ā2
Characteristics of the individual location of anterior circulation aneurysm treated with MPT
| Variables | MCA (%) | ACoA (%) | PCoA (%) |
|---|---|---|---|
| Count | n=256 | n=154 | n=62 |
| Presence of hemorrhage | |||
| ŌĆāUIA | 184 (71.9) | 101 (65.6) | 38 (61.3) |
| ŌĆāRIA | 72 (28.1) | 53 (34.4) | 24 (38.7) |
| Mean size (mm) | 4.67┬▒2.95 | 4.82┬▒2.17 | 4.69┬▒2.25 |
| ŌĆā<5 | 174 (68.0) | 93 (60.4) | 43 (69.4) |
| ŌĆā5-10 | 73 (28.5) | 58 (37.7) | 17 (27.4) |
| ŌĆā>10 | 9 (3.5) | 3 (1.9) | 2 (3.2) |
| Direction of neck | |||
| ŌĆāAnterior | 93 (36.3) | 61 (39.6) | 0 (0.0) |
| ŌĆāPosterior | 30 (11.7) | 17 (11.0) | 54 (87.1)* |
| ŌĆāSuperior | 75 (29.3) | 26 (16.9) | 0 (0.0) |
| ŌĆāInferior | 58 (22.7) | 50 (32.5) | 8 (12.9) |
| Other features (mm) (range) |
 16.52┬▒5.46ŌĆĀ (2.93-30.56) |
 6.52┬▒2.67ŌĆĪ (1.17-14.69) |
 13.15┬▒2.49┬¦ (6.85-18.64) |
Table┬Ā3
Clinical outcomes and complications of 628 patients with 458 MPT and 170 CPT of anterior circulation aneurysms
Table┬Ā4
Survey research to evaluate the functional and cosmetic outcomes of 628 patients with 458 MPT and 170 CPT of anterior circulation aneurysms
- TOOLS
-
METRICS

-
- 3 Crossref
- 0 Scopus
- 4,343 View
- 101 Download
- Related articles




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



