 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 20(3); 2018 > Article |
|
Abstract
Intracranial arterial stenosis usually occurs due to atherosclerosis and is considered the most common cause of stroke worldwide. Although the effectiveness of bypass surgery for ischemic stroke is controversial, the superficial temporal artery to the middle cerebral artery bypass for ischemic stroke is a common procedure. In our report, a 50-year-old man presented with sudden-onset left side weakness and dysarthria. An angiogram showed significant stenosis in the junction of the right cavernous-supraclinoid internal carotid artery and right pericallosal artery. Symptoms altered between improvement and deterioration. Magnetic resonance imaging showed a repeated progression of anterior cerebral artery (ACA) infarction despite maximal medical therapy. We performed a STA-ACA bypass with contralateral STA interposition. Postoperative course was uneventful with no further progression of symptoms. Thus, bypass surgery may be considered in patients with symptomatic stenosis or occlusion of the ACA, especially when patients present progressive symptoms despite maximal medical therapy.
Intracranial arterial stenosis (IAS) is usually attributable to atherosclerosis and corresponds to the most common cause of stroke worldwide.5) S├Īnchez-S├Īnchez et al.17) have found that the anterior cerebral artery (ACA) involvement occurs in 5% of the cases with symptomatic IAS and cerebral infarction in only the ACA territory are not frequent.4)17) Moreover stenosis or occlusion of ACA is often asymptomatic due to ample collateral blood supply. Therefore, symptomatic solitary stenosis or occlusion of ACA is a very rare entity.
Although the effectiveness of bypass surgery for ischemic stroke has remained controversial in previous reports,2)15) the superficial temporal artery (STA) to the middle cerebral artery (MCA) bypass for ischemic stroke is a common procedure, especially when recurrent transient ischemic attack (TIA) or infarction is present despite maximal medical treatment. Several cases of revascularization for symptomatic stenosis or occlusion of ACA have been previously described, yet the effectiveness of ACA revascularization procedures for prevention of strokes is still in question because symptomatic ACA territory infarction due to ACA stenosis or occlusion is a rare clinical situation.7)8)12)15)
In this article, we report our experience of STA to distal ACA bypass with a contralateral STA interposition graft for recurrent progressive symptomatic infarction of ACA territory with stenosis at the A2-A3 junction.
A 50-year-old man with diabetes presented to our emergency room with left homonymous hemianopsia. Brain magnetic resonance imaging (MRI) showed an acute infarction in the territory of the right posterior cerebral artery (PCA) (Fig. 1A). Brain magnetic resonance angiography (MRA) showed stenosis of the right cavernous to the supraclinoid internal carotid artery (ICA) and the right pericallosal artery. A transthoracic echocardiogram and Holter monitoring were performed, but there was no suspicion of cardiogenic embolization. Dual antiplatelet agent was started, and he was discharged without any major neurological deficit.
One month later, he revisited our emergency room, presenting with a tingling sensation in the left arm and decrease of sensation in the left arm and leg. The symptoms started the day before and disappeared immediately after intravenous hydration. MRI showed recent infarctions in the right occipital lobe, right thalamus, and right medial frontal lobe (Fig. 1B). Digital subtraction cerebral angiography (DSA) showed significant stenosis of the right supraclinoid segment of ICA and the right distal ACA (Fig. 1C). Phosphodiesterase inhibitor (cilostazol) was added with dual antiplatelet therapy. Two weeks after discharge, he returned to the emergency room with grade 4 left leg weakness. Diffusion MRI revealed a new infarction of the ACA territory. Symptoms showed repeated improvement and deterioration. MRI performed one week later showed a progression of the ACA infarction. Symptoms were not improved medically; hence, surgical treatment was considered. Single-photon emission computed tomography (SPECT) was performed for preoperative evaluation. Diamox-stressed SPECT showed hypoperfusion at the right ACA, PCA territory with increased perfusion asymmetry, suggesting inadequate vascular reserve function, especially in the ACA territory (Fig. 1D, E).
In the supine position with slight neck flexion, surgical exposure was achieved by a bicoronal incision for the bypass procedure. The bilateral parietal branches of STA were marked with Doppler flowmeter. The left parietal branch of STA was harvested about 7 cm in length for use as interposition graft, and then the right STA was dissected from proximal to distal. After the harvest and donor STA preparation, a right frontal paramedian craniotomy was performed. We identified the appropriate recipient branch of distal ACA in the medial cortex of the frontal hemisphere. Firstly, contralateral STA to ipsilateral STA end to end fashioned anastomosis was done with 10-0 nylon, and then distal ACA to STA bypass end to side fashioned anastomosis was performed. The patency was checked with indocyanine green angiography and Doppler flowmeter (Fig. 2).
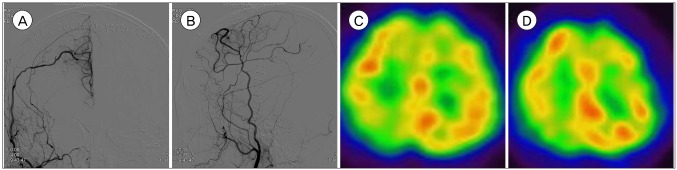
Postoperative course was uneventful with no further progression of symptoms. Postoperative angiograms 4 days after the bypass surgery demonstrated patent STA-STA-ACA anastomosis with perfusion to the right ACA region in the right external carotid artery angiography. The right MCA territory blood supply was observed through the anterior communicating (ACOM) artery and the right posterior communicating (PCOM) artery showed on the left ICA angiography (Fig. 3A, B). Diamox SPECT images obtained at 7 days after surgery showed improved resting perfusion, vascular reserve function of the right ACA territory and no decrease in perfusion in the left ACA territory (Fig. 3C, D). MRI images obtained 6 months later showed no further restriction of diffusion in the brain parenchyma and normal cerebral blood flow in the ACA territory. In addition, the time-to-peak delay was improved compared to previous perfusion images. The patient's clinical symptoms improved, and there was no evidence of symptom recurrence with medication.
The first STA-MCA bypass for treatment of occlusive cerebrovascular disease was performed by Yasargil21) in 1967, and bypass surgery gained popularity as treatment of ischemic stroke. However, since the Extracranial-Intracranial (EC-IC) Bypass Study Group in 1985 failed to prove any benefit of bypass surgery in symptomatic steno-occlusive disease in the MCA or ICA,2) EC-IC bypass as a treatment for intracranial stenosis had rapidly declined.
On the assumption that EC-IC bypass surgery prevents further ischemic stroke in high-risk subgroups of patients with symptomatic major cerebral arterial occlusive diseases, the Japanese EC-IC bypass trial (JET study) was conducted as a prospective randomized multicenter study and demonstrated the effect of EC-IC bypass surgery in preventing subsequent cerebral ischemia in highly selected patients.5) To find evidence of bypass surgery, several modalities were used to evaluate hemodynamic high-risk patients and described hemodynamic changes after bypass surgery. In 1998, a randomized study reported that stage 2 hemodynamic failure (increased oxygen extraction) measured by positron emission tomography) with symptomatic carotid occlusion is an independent prognostic factor for high risk of subsequent stroke despite medical treatment.6) However, discordant with the cerebral hemodynamic evidence in the EC-IC bypass group, EC-IC bypass surgery did not show a decrease in the subsequent stroke rate in The Carotid Occlusion Surgery Study, and previous results failed to be proven in clinical trial.16)
There are different opinions on the interpretation of this result, and the argument that a subgroup of patients with medically refractory symptoms may benefit from bypass surgery has gained support from surgeons.1)3) Currently, bypass surgery is performed for progressive TIA or infarction despite maximal medical therapy. As evidence supporting bypass surgery, several modalities are used to evaluate patients with hemodynamic high risk, and hemodynamic changes are described after bypass surgery. In addition, several studies have followed demonstrating the usefulness of bypass surgery for ischemic stroke.7)12)
The incidence of an ACA territory infarction is reported at 1.1-3% in all cases of strokes.19) Due to the scarcity of the incidence, etiology, mechanism of stroke, and treatment strategy were not clearly defined compared to the infarction of the MCA or PCA territory. Kang et al.11) reported 100 patients with ACA infarction where clinical, MRI, and angiographic findings were evaluated. ACA atherosclerosis was the most frequent etiology of stroke (61%), and among them, in situ thrombo-occlusion or stenosis was the etiology in 20 out of 100 patients (20%).20)
Only a few cases of EC-IC or in situ bypasses for symptomatic occlusion of the ACA have been reported, and established treatments concerning the effectiveness of bypass technique for the prevention of infarctions remains controversial because of its rarity. The previously reported cases of ACA bypass for ischemic steno-occlusion of ACA are listed and described in Table 1.
A3-A3 side to side in situ bypass technique has been widely used for revascularization of complex ACA aneurysms in the literature.8) Ikeda et al.8) reported two cases where inter-ACA anastomoses were performed for symptomatic occlusion of ACA with poor collateral. Another report10) described an A3-A3 in situ bypass which was performed for unilateral ACA stenosis or occlusion. The A3-A3 bypass has the advantage of not requiring graft harvesting and additional surgical incision. However, deep sutures are needed which require technical expertise, and the most problematic issue of the A3-A3 in-situ bypass is nearly always the decreased flow of the normal side due to the steal phenomenon.8) In our case, the steal phenomenon was expected if ACA flow was increased with the bypass, since pre-operative angiogram showed right supraclinoid-cavernous segment stenosis, and blood flow to the right MCA territory was supplied from the left ICA through the ACOM and PCOM artery.
As the other bypass option, EC-IC (STA-ACA) bypass has been used to flow augmentation. The ipsilateral parietal branch of STA to cortical ACA bypass in unilateral ACA symptomatic stenosis was reported by Lee et al.13) However, the length and caliber of the parietal branch of the STA were not sufficient for direct ACA to STA anastomosis in many cases. Therefore, radial artery,12) occipital artery,14) cephalic vein,9) and contralateral STA14) free grafts were used for interposition graft. Horiuchi et al.7) reported on the STA-STA-ACA bypass combined with A3-A3 bypass which was performed in patients with bilateral stenosis or occlusion of the ACA which was done to reduce the risk of watershed zone infarction and steal phenomenon. In our case, we thought that caliber and length of the parietal branch of STA in preoperative extracranial angiography was not enough for augmentation of ACA flow. In addition, STA-ACA interposition graft procedure does not require the additional remote surgical graft harvesting procedure which also may cause related complications, such as numbness, pain and other problems in the graft site. Since our case is a unilateral lesion with the possibility of steal phenomenon, we concluded that interposition graft using contralateral STA free graft without additional A3-A3 in situ bypass was the optimal procedure for the patient.
Bypass surgery may be considered for patients with symptomatic stenosis or occlusion of the ACA, especially in patients with progressive symptoms despite maximal medical therapy. Contralateral STA interposition graft can be used as a good surgical option when the length of the STA and the caliber of the donor artery is not enough.
References
1. Amin-Hanjani S, Barker FG 2nd, Charbel FT, Connolly ES Jr, Morcos JJ, Thompson BG, et al. Extracranial-intracranial bypass for stroke-is this the end of the line or a bump in the road? Neurosurgery. 2012 9;71(3):557-561;



2. Baron JC, Bousser MG, Rey A, Guillard A, Comar D, Castaigne P. Reversal of focal ŌĆ£misery-perfusion syndromeŌĆØ by extra-intracranial arterial bypass in hemodynamic cerebral ischemia. A case study with 15O positron emission tomography. Stroke. 1981 Jul-Aug;12(4):454-459;


3. Carlson AP, Yonas H, Chang YF, Nemoto EM. Failure of cerebral hemodynamic selection in general or of specific positron emission tomography methodology?: Carotid Occlusion Surgery Study (COSS). Stroke. 2011 12;42(12):3637-3639;



4. Carvalho M, Oliveira A, Azevedo E, Bastos-Leite AJ. Intracranial arterial stenosis. J Stroke Cerebrovasc Dis. 2014 4;23(4):599-609;


6. Grubb RL Jr, Derdeyn CP, Fritsch SM, Carpenter DA, Yundt KD, Videen TO, et al. Importance of hemodynamic factors in the prognosis of symptomatic carotid occlusion. JAMA. 1998 9;280(12):1055-1060;


7. Horiuchi T, Ichinose S, Agata M, Ito K, Hongo K. STA-ACA bypass using the ipsilateral free STA graft as an interposition graft and A3-A3 anastomosis for treatment of bilateral ACA steno-occlusive ischemia. Acta Neurochir (Wien). 018 4;160(4):779-782;


8. Ikeda A, Okada T, Shibuya M, Noda S, Sugiura M, Iguchi I, et al. Revascularization of the anterior cerebral artery. Report of two cases. J Neurosurg. 1985 4;62(4):603-606;


9. Ishii R, Koike T, Takeuchi S, Ohsugi S, Tanaka R, Konno K. Anastomosis of the superficial temporal artery to the distal anterior cerebral artery with interposed cephalic vein graft. Case report. J Neurosurg. 1983 3;58(3):425-429;


10. Ito Z. A new technique of intracranial interarterial anastomosis between distal anterior cerebral arteries (ACA) for ACA occlusion and its indication. Neurol Med Chir (Tokyo). 1981 9;21(9):931-939;


11. Kang SY, Kim JS. Anterior cerebral artery infarction: Stroke mechanism and clinical-imaging study in 100 patients. Neurology. 2008 6 10 70:2386


12. Kiyofuji S, Inoue T, Hasegawa H, Tamura A, Saito I. A3-A3 anastomosis and superficial temporal artery-radial artery graft-A3 bypass to treat bilateral ACA steno-occlusive hemodynamic ischemia with cognitive and executive dysfunction: a technical note. Acta Neurochir (Wien). 2014 11;156(11):2085-2093; discussion 2093.



13. Lee SC, Ahn JH, Kang HS, Kim JE. Revascularization for symptomatic occlusion of the anterior cerebral artery using superficial temporal artery. J Korean Neurosurg Soc. 2013 12;54(6):511-514;




14. Nagm A, Horiuchi T, Yanagawa T, Hongo K. Risky Cerebrovascular anatomic orientation: implications for brain revascularization. World Neurosurg. 2016 12;96:610.e15-610.e20.


15. Park ES, Ahn JS, Park JC, Kwon DH, Kwun BD, Kim CJ. STA-ACA bypass using the contralateral STA as an interposition graft for the treatment of complex ACA aneurysms: report of two cases and a review of the literature. Acta Neurochir (Wien). 2012 8;154(8):1447-1453;



16. Powers WJ, Clarke WR, Grubb RL Jr, Videen TO, Adams HP Jr, Derdeyn CP, et al. Extracranial-intracranial bypass surgery for stroke prevention in hemodynamic cerebral ischemia: the Carotid Occlusion Surgery Study randomized trial. JAMA. 2011 11;306(18):1983-1992;



17. S├Īnchez-S├Īnchez C, Egido JA, Gonz├Īlez-Guti├®rrez JL, Mera-Campillo J, Carneado-Ruiz J, D├Łaz-Otero F. Stroke and intracranial stenosis: clinical profile in a series of 134 patients in Spain. Rev Neurol. 2004 8;39(4):305-311;

18. Terasaka S, Satoh M, Echizenya K, Murai H, Fujimoto S, Asaoka K. Revascularization of the anterior cerebral artery using a free superficial temporal artery graft: a case report. Surg Neurol. 1997 8;48(2):164-169; discussion 169-70.


19. Vemmos KN, Takis CE, Georgilis K, Zakopoulos NA, Lekakis JP, Papamichael CM, et al. The Athens stroke registry: results of a five-year hospital-based study. Cerebrovasc Dis. 2000 Mar-Apr;10(2):133-141;


20. Wityk RJ, Lehman D, Klag M, Coresh J, Ahn H, Litt B. Race and sex differences in the distribution of cerebral atherosclerosis. Stroke. 1996 11;27(11):1974-1980;


21. Yasargil MG. Experimental small vessel surgery in the dog including patching and grafting of cerebral vessels and the formation of functional extra-intracranial shunts. In: Donaghy RMP, Yasargil MG, editors. Micro-vascular Surgery. Stuttgart: Georg Thieme Verlag; 1967. p. 87-126.
Fig.┬Ā1
Preoperative MRI, cerebral angiography, and SPECT. (A) Initial MRI shows acute infarction on the territory of right ACA. (B) 1-month follow up MRI shows further progression of the ACA infarction. (C) Cerebral angiography reveals right supraclinoid cavernous stenosis and right distal ACA (white arrow). (D) Basal and (E) Diamox SPECT shows decreased vascular reserve function in the ACA territory. MRI = magnetic resonance imaging; SPECT = single-photon emission computed tomography; ACA = anterior cerebral artery.
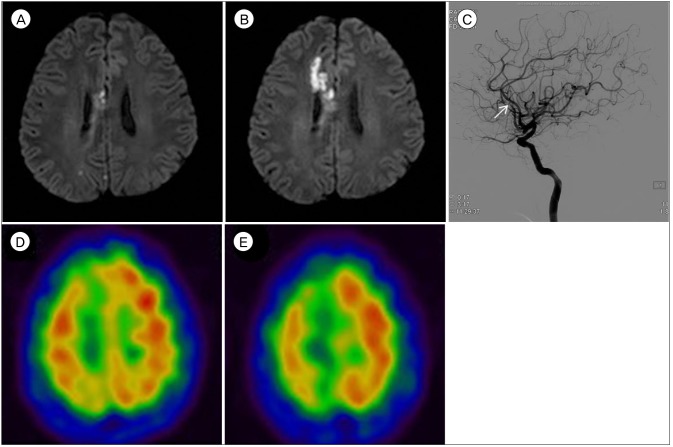
Fig.┬Ā2
Intraoperative photograph and ICG angiography. (A) Contralateral STA to ipsilateral STA end to end fashioned anastomosis and (B) distal ACA to STA bypass end to side fashioned anastomosis were performed. (C) Intraoperative ICG angiography. ICG = indocyanine green; STA = superficial temporal artery; ACA = anterior cerebral artery.

Fig.┬Ā3
Cerebral angiography obtained 4 days after surgery and perfusion MRI at 6 months after surgery. (A, B) Blood flows to the right ACA region from right ECA. (C) Basal and (D) Diamox SPECT show improved perfusion status, compared to previous SPECT. MRI = magnetic resonance imaging; ACA = anterior cerebral artery; SPECT = single-photon emission computed tomography.
Table┬Ā1
The previously reported cases of ACA bypass for ischemic steno-occlusion of ACA
| Study | Sex/age | Stenosis location | Clinical presentation | Bypass vessles | Anatomosis | Craniotomy | Graft |
|---|---|---|---|---|---|---|---|
| Ishii et al9) (1983) | F/30 | A1 | Left arm weakness | STA-Cephalic vein-A4 (callosomarginal) | End to end | Bifrontal and right temporoparietal craniotomy | Cephalic vein |
| End to side | |||||||
| Ikeda et al8) (1985) | M/50 | A2 | Right leg weakness | A3 to A4 | Side to side | paramedian craniotomy | |
| M/52 | Right ICA ACOM | Left side weakness | 1. STA-MCA | - | |||
| 2. PIFA-PIFA | Side to side | ||||||
| Terasaka et al18) (1997) | M/38 | A2-A3 junction | Left leg weakness | A2-STA-A3 | Side to end | Bifrontal craniotomy | STA |
| End to side | |||||||
| Lee et al13) (2013) | M/69 | A2 | Left leg weakness | STA-distal ACA | End to Side | Paramedian craniotomy | |
| Kiyofuji et al12) (2014) | F/74 | A2 | Left side weakness | A3-A3-RA-STA | Side to side | Bicoronal | Radial artery |
| Side to end | |||||||
| End to end | |||||||
| F/73 | A2 | Left leg weakness | As above | As above | As above | As above | |
| Nagm et al14) (2016) | F/71 | A2 | Right leg weakness | STA-OA-PIFA | End to end | Medial frontal craniotomy | OA |
| End to side | |||||||
| Horiuchi et al7) (2018) | F/71 | A2 | Left side weakness | ACA-OA-STA | - | - | OA |
| M/50 | A2 | Left side weakness | A3-A3 | - | - | ||
| F/70 | A1 | Right leg weakness | A3-A3-STA-STA | Side to side | Paramedian craniotomy | STA | |
| Side to end | |||||||
| End to end |
- TOOLS
-
METRICS

-
- 5 Crossref
- 0 Scopus
- 2,628 View
- 27 Download
- Related articles




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



