 |
 |
| J Cerebrovasc Endovasc Neurosurg > Volume 20(2); 2018 > Article |
|
Abstract
Objective
In our series, endovascular coiling with Target┬« NanoŌäó coils (Stryker Neurovascular, Fremont, CA, USA) with diameters of 1 or 1.5 mm exhibited favorable technical feasibility in the treatment of small cerebral aneurysms (< 4 mm). However, little is known about the recurrence of small cerebral aneurysms treated using Target┬« NanoŌäó coils. We investigated recurrence following the treatment of small cerebral aneurysms using Target┬« NanoŌäó coils.
Materials and Methods
Between January 2012 and November 2013, 143 patients with 148 small cerebral aneurysms (< 4 mm) were included our study. A total of 135 cerebral aneurysms (91.2%) were unruptured; 45 cerebral aneurysms (30.4%) were treated by endovascular coiling using Target┬« NanoŌäó coils. Follow-up radiological images were obtained for 132 cerebral aneurysms (89.2%) over a range of 3 to 58 months (mean, 34.3 months; standard deviation, 14.2).
Results
In the group treated with Target┬« NanoŌäó coils, radiological outcomes revealed complete occlusion in 33 (73.3%), residual necks in eight (17.8%), and residual sacs in four (8.9%) cases. Follow-up radiological outcomes revealed complete occlusion in 35 (77.8%) and residual necks in four (8.9%) cases that exhibited stable coil masses. In the group that was not treated with Target┬« NanoŌäó coils, radiological outcomes revealed complete occlusion in 69 (67%), residual necks in 18 (17.5%), and residual sacs in 16 (15.5%) cases. Follow-up radiological outcomes revealed complete occlusion in 87 (84.5%) and residual necks (5.8%) in six cases that exhibited stable coil masses. No significant differences were observed in the radiological outcomes or follow-up radiological outcomes between the two groups. No recurrences or retreatments occurred in our series.
Recently, Target┬« NanoŌäó coils (Stryker Neurovascular, Fremont, CA, USA) have increasingly been used to treat small cerebral aneurysms and have demonstrated favorable technical feasibility and relatively high coil compaction densities.5)6)14) Theoretically, a higher coil packing density may be an influential factor in terms of reducing recurrence. However, little is known about the recurrence of small cerebral aneurysms treated with Target┬« NanoŌäó coils. Therefore, we retrospectively investigated the recurrence of small cerebral aneurysms (< 4 mm) endovascularly treated using Target┬« NanoŌäó coils.
Between January 2012 and November 2013, 143 patients with 148 small cerebral aneurysms (< 4 mm) who underwent endovascular treatment in two hospitals were included in our study. The study was approved by the Institutional Review Board of our institution, and all subjects provided informed consent.
Our series included 117 female patients (79.1%) with a mean age of 56.6 years (range, 31 to 82 years), and 135 of the cerebral aneurysms (91.2%) were unruptured. The locations of the aneurysms were in the paraclinoid region (63, 42.6%), posterior communicating artery segments (27, 18.3%), anterior communicating arteries (23, 15.5%), proximal anterior cerebral artery (1, 0.7%), middle cerebral artery bifurcation (11, 7.4%), proximal middle cerebral arteries (5, 3.4%), internal cerebral artery bifurcation (3, 2.0%), dorsal wall of the internal cerebral artery (3, 2.0%), basilar bifurcation (3, 2.0%), basilar trunk (2, 1.4%), posterior inferior cerebellar arteries (2, 1.4%), and superior cerebellar arteries (2, 1.4%). No significant differences were observed in the demographic or aneurysmal characteristics between the groups treated with or without the Target┬« NanoŌäó coils (the diameters of the coils were 1 or 1.5 mm).7) The endovascular modalities in our series were as follows: 68 single catheter (45.9%) procedures, 10 double microcatheter (6.8%) procedures, 69 stent-assisted (46.6%) procedures, and 1 wire-assisted (0.7%) procedure. A total of 45 (30.4%) cerebral aneurysms were treated endovascularly with Target┬« NanoŌäó coils.
In our practice, ruptured small cerebral aneurysms (< 4 mm) are selectively treated with endovascular coiling on an emergent basis according to the selection of the treatment modality. For small unruptured cerebral aneurysms, and particularly in cases of bifurcated aneurysms or posterior circulation aneurysms, we recommend treatment if the patient is 70 years old or younger. For small cerebral aneurysms in other locations, the treatment decision should be based on the morphology and multiplicity of the aneurysm(s), previous history of subarachnoid hemorrhage (SAH), the age and emotional status of the patient, and/or technical feasibility. In cases of cerebral aneurysms smaller than 2 mm, annual follow-up evaluations are generally recommended.
The clinical data were obtained by reviewing the patients' medical records. Using procedural records and images, the characteristics of the aneurysms, including the location, size (width, depth, and height), neck diameter, vessel incorporation and procedural details, such as the immediate angiographic results and complications, were reviewed. Follow-up clinical outcomes were defined according to the modified Rankin Scale (mRS).2)9) Good clinical follow-up outcomes were defined as mRS scores of 0 or 1.
A simple coiling method using single or multiple microcatheters was generally used. The multiple microcatheter technique was applied to aneurysms with broad necks or incorporated vessels according to technical details that have been previously described.7)8) Stent-assisted coiling or balloon-assisted coiling was performed for small cerebral aneurysms that were not suitable for simple coiling or following a failed attempt at simple coiling according to the interventionist's discretion. All procedures were performed under general anesthesia using two biplane systems at two institutions (IntegrisAllura, Phillips Medical Systems, CITY, the Netherlands and Artis ZEE, Siemens, Germany).
The angiographic findings, including the aneurysm size and degree of occlusion, were interpreted by neurointerventional neurosurgeons or radiologists. Aneurysm size was measured based on 3-dimensional angiographic images. Aneurysm width was determined by measuring the longest diameter of the fundus parallel to the axis of the aneurysm neck. The height was determined as the longest diameter of the fundus vertical to the axis of the aneurysm neck. Aneurysm depth was determined by measuring the longest diameter of an aneurysm perpendicular to the axis of the parental artery. Aneurysm volume was calculated by assuming that the aneurysms were elliptical and using the following formula: aneurysm volume = 4 ŽĆ (height / 2 ├Ś length / 2 ├Ś width / 2) / 3.
The coil volumes were calculated using the following formula: coil volume = ŽĆ (radius)2 ├Ś length.
The coil packing density was calculated using the following formula: packing density = (coil volume / aneurysm volume) ├Ś 100%.
The angiographic outcomes were classified as complete occlusion, residual neck, and partial occlusion according to the Raymond scale.8)
Statistical analyses were performed to evaluate the differences in the follow-up radiological outcomes of small cerebral aneurysms according to the use of Target┬« NanoŌäó coils. Nominal data were analyzed using the Žć2 or Fisher exact tests, and numerical data were analyzed using Student's t-tests or Mann-Whitney U tests as appropriate. A two-tailed value of p < 0.05 was defined as statistically significant.
No procedural failures occurred in the endovascular coiling of small cerebral aneurysms (< 4 mm) with or without the Target┬« NanoŌäó coils. The radiological outcomes revealed complete occlusion in 102 cases (68.9%), a residual neck in 26 cases (17.6%), and a residual sac in 20 cases (13.5%) (Fig. 1, 2). No significant differences were observed in the endovascular techniques or immediate radiological outcomes between the two groups.6) Follow-up radiological evaluations, such as brain magnetic resonance angiography (MRA) or conventional angiography, were possible for 132 of the cerebral aneurysms (89.2%). The mean radiological follow-up time was 34.3 months with a range of 3 to 58 months. In the Target┬« NanoŌäó coil group (n = 45), follow-up radiological evaluations were possible for 39 lesions (86.7%). Of these, 35 lesions (89.7%) exhibited complete occlusion, residual necks were present in four lesions (10.3%), and all exhibited stable coil masses. In the non-Target┬« NanoŌäó coil group (n = 103), follow-up radiological evaluations were available for 93 lesions (90.3%). Of these, complete occlusion was present in 87 lesions (93.5%), residual necks were present in six lesions (6.5%), and all exhibited stable coil masses. No significant difference was observed in the follow-up radiological outcomes between the two groups (p = 0.481) (Table 1).
Follow-up clinical evaluations over a range of 2 to 65 months (mean, 39.5 months; standard deviation, 13.74) were possible for 140 lesions (94.6%). In the Target┬« NanoŌäó coil group, follow-up clinical data were available for 43 lesions (95.6%), and the mRS scores were 0 for 41 lesions, 2 for 1 lesion, and 4 for 1 lesion. The two lesions with mRS scores Ōēź 2 had initial clinical statuses of ruptured cerebral aneurysms. In the group treated without Target┬« NanoŌäó coils, follow-up clinical data were available for 97 (94.2%) lesions, and the mRS scores were 0 for 92 lesions, 1 for 2 lesions, 2 for 2 lesions, and 4 for 1 lesion. The unruptured cerebral aneurysms exhibited good follow-up clinical outcomes. The lesions with scores Ōēź 2 were ruptured cerebral aneurysms, and both unruptured and ruptured cerebral aneurysms were correlated with the initial clinical status. In our series, no ruptures of the aneurysms were observed during follow-up.
In general, endovascular coiling provides favorable clinical outcomes and a relatively low procedural morbidity. However, the durability, as described by recurrence, is a major drawback of endovascular coiling.11)12)15) For small cerebral aneurysms, it was recently reported that endovascular coiling using Target┬« NanoŌäó coils results in higher packing density and favorable technical feasibility. However, although higher packing density may be associated with a reduction in recurrence, higher packing density is not as important as low recurrence. Therefore, recurrence after the endovascular coiling of small cerebral aneurysms using Target┬« NanoŌäó coils has rarely been reported and should be evaluated.
Because there is no enough space for microcatheter or microwire to be navigated into the small cerebral aneurysms, aneurysmal rupture could be the most fatal complication during endovascular coiling for small cerebral aneurysms. In our series, there was no procedural aneurysm rupture in endovascular coiling of small cerebral aneurysms, using NanoŌäó coils. To prevent procedural aneurysm rupture during endovascular coiling of small cerebral aneurysms, adequate shaping of distal microcatheter tip should be prepared to easily navigate into the aneurysm to avoid struggling of superselection. Also, further push of microcatheter should also be avoided when trying to modulate tension of microcatheter.
Theoretically, the uneven packing of the compartment of a cerebral aneurysm with coils could be reduced by decreasing the coil diameter compared to the size of the aneurysm. Loose coil packing may be related to continuous blood flow into the aneurysmal sac and could increase the recurrence rate following endovascular coiling.1)3)10) The group that underwent Target┬« NanoŌäó coil packing generally had a smaller coil diameter to compartment ratio than the non-nano coil group, which could be expected to result in a higher recurrence or retreatment rate in the group using Target┬« NanoŌäó coils. However, in our series, no significant differences were observed in the radiological outcomes of the groups treated with and without Target┬« NanoŌäó coils. No recurrences or retreatments occurred in either group. Based on our series, compartmentalization of the aneurysm by coil packing is not a critical factor in terms of the recurrence of small cerebral aneurysms (< 4 mm). Compartment packing with coils in small cerebral aneurysms (< 4 mm) may result in the stagnation of blood flow rather than continuous filling of the aneurysm with blood and thus might be related to spontaneous thrombosis of the aneurysmal sac portion due to loosely packed coils. Incomplete occlusion, including residual sacs or necks, and subsequent complete obliteration of the residual aneurysm on follow-up angiograms have been reported following the endovascular coiling of very small cerebral aneurysms and paraclinoid sidewall aneurysms,1)4) and situations that could be caused by spontaneous thrombosis of the residual or compartmentalized aneurysm sac may support our results. Because the proportion of ruptured cerebral aneurysms (i.e., a major risk factor for recurrence after endovascular coiling) was small in our series (8.8%), our suggestion that endovascular coiling using Target┬« NanoŌäó coils is a durable treatment method cannot be accepted for ruptured small cerebral aneurysms (< 4 mm). This is one of the limitations of our study. Our series also has the limitation of being a retrospective, non-randomized, and small sample-size study. Nevertheless, our study may have clinical value and indicates that endovascular coiling using Target┬« NanoŌäó coils may be a durable treatment option for unruptured small cerebral aneurysms (< 4 mm).
References
1. Bavinzski G, Talazoglu V, Killer M, Richling B, Gruber A, Gross CE, et al. Gross and microscopic histopathological findings in aneurysms of the human brain treated with guglielmi detachable coils. J Neurosurg. 1999 8;91(2):284-293;


2. Biondi A, Janardhan V, Katz JM, Salvaggio K, Riina HA, Gobin YP. Neuroform stent-assisted coil embolization of wide-neck intracranial aneurysms: strategies in stent deployment and midterm follow-up. Neurosurgery. 2007 9;61(3):460-468;



3. Brinjikji W, Kallmes DF, Kadirvel R. Mechanisms of healing in coiled intracranial aneurysms: a review of the literature. AJNR Am J Neuroradiol. 2015 7;36(7):1216-1222;



4. Hayakawa M, Murayama Y, Duckwiler GR, Gobin YP, Guglielmi G, Vi├▒uela F. Natural history of the neck remnant of a cerebral aneurysm treated with the guglielmi detachable coil system. J Neurosurg. 2000 10;93(4):561-568;


5. Ioannidis I, Lalloo S, Corkill R, Kuker W, Byrne JV. Endovascular treatment of very small intracranial aneurysms. J Neurosurg. 2010 3;112(3):551-556;


6. Jeong HW, Jin SC. Practical feasibility and packing density of endovascular coiling using target┬« nanoŌäó coils in small cerebral aneurysms. J Cerebrovasc Endovasc Neurosurg. 2015 12;17(4):295-300;



7. Kwon OK, Kim SH, Kwon BJ, Kang HS, Kim JH, Oh CW, et al. Endovascular treatment of wide-necked aneurysms by using two microcatheters: techniques and outcomes in 25 patients. AJNR Am J Neuroradiol. 2005 4;26(4):894-900;


8. Kwon OK, Kim SH, Oh CW, Han MH, Kang HS, Kwon BJ, et al. Embolization of wide-necked aneurysms with using three or more microcatheters. Acta Neurochir (Wien). 2006 11;148(11):1139-1145; discussion 1145.



9. Molyneux AJ, Kerr RS, Birks J, Ramzi N, Yarnold J, Sneade M, et al. Risk of recurrent subarachnoid haemorrhage, death, or dependence and standardised mortality ratios after clipping or coiling of an intracranial aneurysm in the International Subarachnoid Aneurysm Trial (ISAT): long-term follow-up. Lancet Neurol. 2009 5;8(5):427-433;



10. Murayama Y, Nien YL, Duckwiler G, Gobin YP, Jahan R, Frazee J, et al. Guglielmi detachable coil embolization of cerebral aneurysms: 11 years' experience. J Neurosurg. 2003 5;98(5):959-966;


11. Rabinstein AA, Nichols DA. Endovascular coil embolization of cerebral aneurysm remnants after incomplete surgical obliteration. Stroke. 2002 7;33(7):1809-1815;


12. Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, Lambert A, et al. Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke. 2003 6;34(6):1398-1403;


Fig.┬Ā1
Left SCA unruptured aneurysm (2.3 ├Ś 3.1 mm) of 63 year old female. Pre-procedural images of Angiography, white arrow (A) and three dimensionally reconstructed image (B) are shown. Aneurysm is being treated via wire assisted embolization (C, D) and partially coiled (E). 30 months follow up brain MR angiography shows complete embolization, red arrow (F).
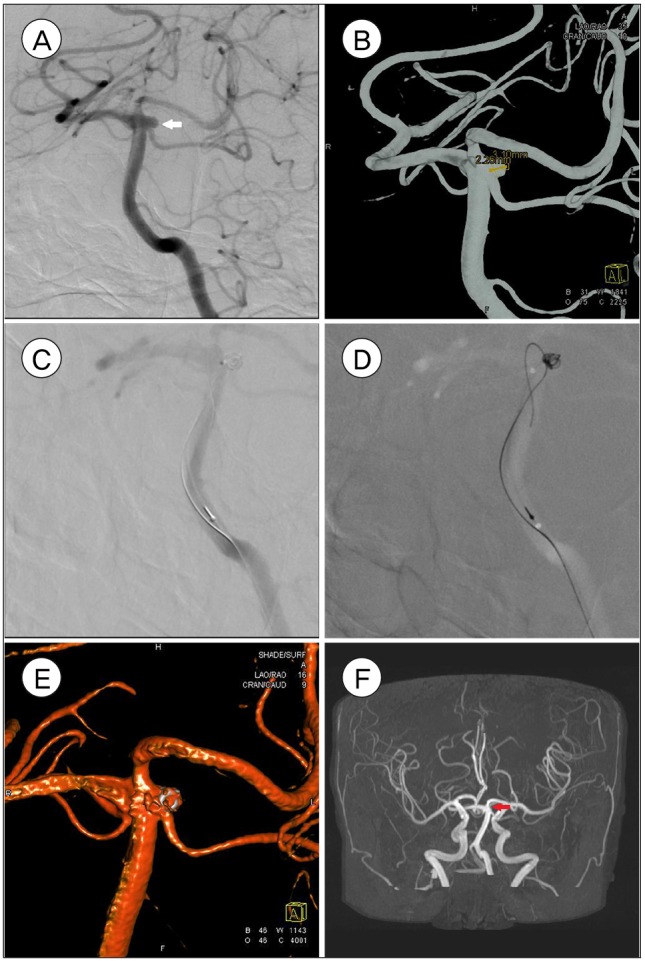
Fig.┬Ā2
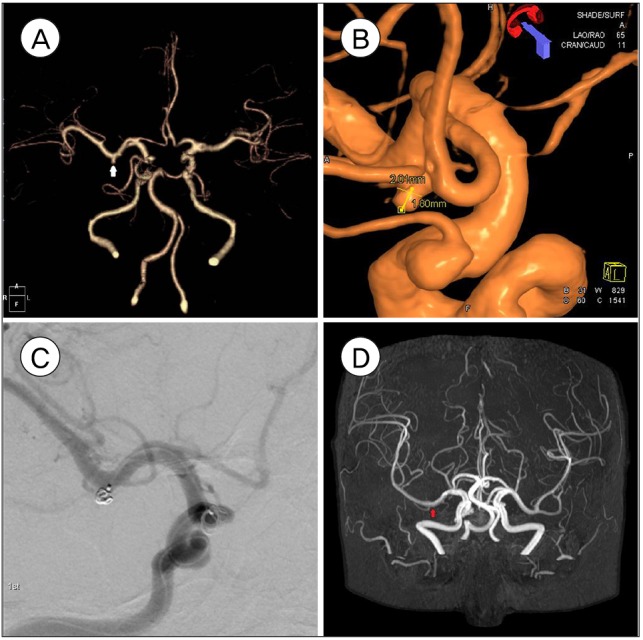
Right M1 unruptured aneurysm (2.0 ├Ś 1.8 mm) of 76 year of female. Pre-procedural images of computed tomography angiogram, white arrow (A) and three dimensionally reconstructed angiography (B) are shown. Aneurysm is being treated with partial embolization (C) and 4.5 years follow up brain magnetic resonance angiography shows complete embolization, red arrow (D).
Table┬Ā1
Comparison of coil technique, radiological outcome, and followed radiological outcome between small cerebral aneurysms treated with NanoTM and without NanoŌäó coils
- TOOLS
-
METRICS

-
- 2 Crossref
- 0 Scopus
- 2,507 View
- 35 Download
- Related articles
-
Fusiform Superior Cerebellar Artery Aneurysm Treated with Endovascular Treatment2016 September;18(3)




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



